The Case for Combining HRT and GLP-1s: A New Framework for Menopausal Healthspan
Prefer to listen? Hit play for a conversational, audio‑style summary of this article’s key points.
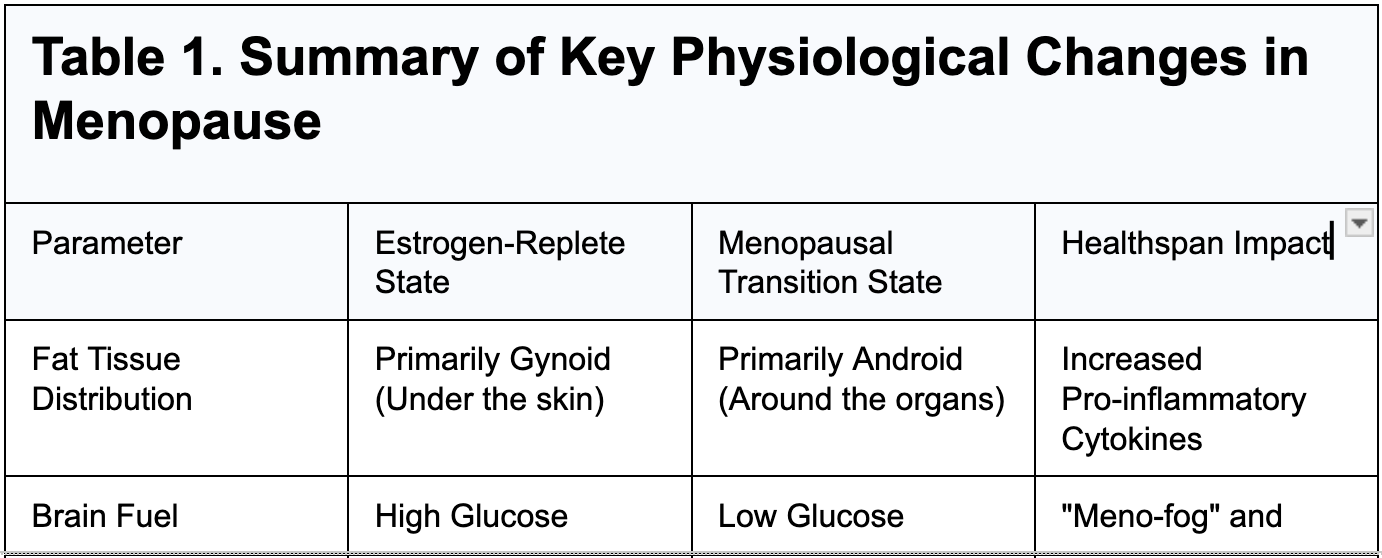
Menopause is a coordinated systemic disruption, not a collection of isolated symptoms. The decline of estrogen and progesterone withdraws hormonal signals that had been governing cellular energy regulation, mitochondrial function, and inflammatory tone across the brain, vasculature, adipose tissue, and skeletal muscle simultaneously. Treating it symptom by symptom produces, at best, partial solutions.
The brain is one of the earliest and most consequential targets of estrogen withdrawal. As estrogen declines, neuronal glucose metabolism becomes measurably impaired, creating an energetic deficit that manifests as cognitive fatigue and memory disruption. Sustained over years, this deficit has been linked to the early biological processes associated with Alzheimer's disease, which affects women at roughly twice the rate of men.
Menopause accelerates visceral fat accumulation and worsens insulin sensitivity through distinct biological mechanisms. Visceral fat actively secretes pro-inflammatory cytokines including IL-6 and hsCRP, creating chronic low-grade inflammation that progressively impairs metabolic health and elevates risk for type 2 diabetes, cardiovascular disease, and metabolic syndrome.
The Women's Health Initiative findings that shaped two decades of HRT avoidance were highly context-dependent. The average participant was 63 and more than a decade past menopause. The timing hypothesis now established in the literature suggests that initiating HRT before age 60 or within 10 years of menopause is associated with a substantially more favorable risk profile and, in some analyses, active cardiovascular benefit.
HRT and GLP-1 receptor agonists address different levels of the same biological disruption. HRT acts upstream, restoring the hormonal signaling environment that menopause withdraws. GLP-1 receptor agonists act downstream, targeting the visceral fat accumulation, insulin resistance, and dysregulated appetite that estrogen's absence creates. They are complementary tools, not alternatives.
Both therapies converge on the same cellular signaling pathways, and their combined effect may be greater than the sum of their parts. Estrogen and GLP-1 receptor agonists both activate PI3K-Akt, which governs insulin sensitivity and cell survival, and AMPK, the cell's master energy sensor. Their shared engagement of these pathways may produce a more integrated physiological response than either achieves independently.
The first human data on the combination are early but striking. A 2026 retrospective study in The Lancet found that postmenopausal women receiving tirzepatide alongside hormone therapy were more than four times as likely to achieve 30% weight loss compared to those on tirzepatide alone: 18% versus 4%. At 20% weight loss, the figures were 45% versus 24%.
Up to 25 to 40% of weight lost during GLP-1 therapy may come from lean muscle mass if not actively addressed. For menopausal women already navigating sarcopenia, this carries significant long-term consequences. Muscle is the primary site of insulin-mediated glucose disposal, meaning losing it worsens the very insulin resistance GLP-1 therapy is designed to improve.
Formulation and route of delivery are not interchangeable and carry real clinical consequences. Transdermal estradiol carries meaningfully lower thrombotic risk than oral estrogen. Micronized progesterone demonstrates a more favorable cardiometabolic and neurocognitive profile than older synthetic progestins. These are strategic decisions that either align with or work against the broader healthspan objectives of the combined approach.
The neuroprotective potential of this combination may prove to be its most significant long-term benefit. Preclinical research on GLP-1 and estrogen conjugates demonstrates enhanced autophagy for clearing amyloid and tau aggregates, reduced neuroinflammation, and improved synaptic function. If these mechanisms translate to humans, combined hormonal and metabolic strategies may represent a meaningful new direction in preserving cognitive function during aging.
Reframing Menopause for Long-Term Health
Each year, millions of women pass through one of the most consequential biological transitions of their lives, yet it remains one of the least studied and most poorly understood in mainstream medicine. Menopause, affecting an estimated 1.55 billion women worldwide, is still frequently framed as a reproductive endpoint, a cessation of fertility marked by the end of menstrual cycles. That framing, while not wrong, misses most of what is actually happening.
Menopause is not simply the body winding down one system. It is a systemic physiological reorganization, one that ripples outward from the ovaries into metabolism, body composition, cardiovascular function, bone density, and cognitive resilience. The hormones that decline during this transition, primarily estrogen and progesterone, are not merely reproductive signals. They are metabolic regulators, operating across virtually every tissue in the body. When their levels fall, the consequences extend far beyond the symptoms most commonly associated with menopause: hot flashes, disrupted sleep, and mood changes. These visible symptoms are, in many ways, the surface expression of a much deeper shift in how cells produce and manage energy.
Historically, clinical care has responded to that surface. Symptoms have been treated in isolation, hot flashes managed here, bone loss addressed there, mood changes handled separately. What has been slower to emerge is a framework that recognizes these not as independent problems but as coordinated consequences of a single underlying disruption: the withdrawal of hormonal signals that had, for decades, been quietly governing cellular energy regulation, inflammatory tone, and tissue maintenance throughout the body.
That recognition is now reshaping how researchers and clinicians think about the menopausal transition. Two therapeutic approaches in particular are drawing increasing attention, not as standalone symptom managers but as potential tools for addressing the deeper biology of this transition. Hormone replacement therapy, or HRT, works by restoring the hormonal signaling that menopause withdraws, supporting the mitochondrial function, tissue health, and metabolic stability that estrogen had been maintaining. GLP-1 receptor agonists, a class of drugs originally developed for type 2 diabetes and obesity that have recently transformed metabolic medicine, target the downstream consequences of that hormonal withdrawal: dysregulated energy metabolism, inflammation, and shifts in body composition. [1]
Used together, these two approaches may offer something that neither provides alone: a framework for addressing both the hormonal drivers and the metabolic consequences of menopause simultaneously. Understanding why that combination is compelling requires first understanding what menopause actually does to the body at the cellular level, and why the consequences are so far-reaching, extending well beyond the symptoms that have historically defined how this transition is understood and treated.
The Bioenergetic Crisis of the Menopause Transition
The hormonal shifts of menopause are often described in terms of what is lost: estrogen falls, progesterone declines, and the reproductive system transitions out of its active phase. But framing menopause primarily as a reproductive event obscures what is biologically more significant. Estrogen and progesterone are not narrow-purpose hormones. They are systemic regulators, operating across the brain, cardiovascular system, skeletal muscle, bone, and adipose tissue simultaneously. When their levels fall, the consequences cascade outward into nearly every domain of physiology.
Understanding why requires stepping back from the symptom level and looking at what these hormones were actually doing at the cellular level all along.
The Brain's Energy Crisis
Among the least discussed but most consequential effects of estrogen withdrawal is what happens inside the brain. Estrogen plays an active role in neuronal glucose metabolism and mitochondrial bioenergetics, the processes by which brain cells generate the energy required to function. It does this partly by facilitating the localization of key metabolic enzymes within mitochondria, supporting their structural integrity and protecting against oxidative damage to mitochondrial DNA [2]. In practical terms, estrogen helps keep the brain's energy production running efficiently.
As estrogen levels decline, that support is withdrawn. The brain's ability to utilize glucose becomes measurably impaired, creating an energetic deficit at the neuronal level. Clinically, this often surfaces as cognitive fatigue, difficulty with memory retrieval, and the phenomenon that many women describe as "menofog," a pervasive sense of mental cloudiness that is not simply tiredness but reflects a genuine disruption in how brain cells are producing and managing energy [2].
The longer-term implications are more sobering. Sustained deficits in neuronal energy metabolism have been linked to the early stages of neurodegenerative processes, including the accumulation of beta-amyloid plaques and tau protein tangles, both hallmarks of Alzheimer's disease pathology [2]. This has led researchers to characterize the menopausal transition as a sex-specific risk window, a period during which the metabolic and neurobiological changes set in motion by hormonal withdrawal may meaningfully shape long-term disease trajectories [3]. Alzheimer's disease affects women at roughly twice the rate of men, and the hormonal biology of menopause is increasingly understood as a contributing factor to that disparity, not merely a coincidence of longer average lifespan [3].
A Shift in Where the Body Stores Fat
The brain is not the only system reorganizing during this transition. Systemic metabolism undergoes significant structural changes as estrogen withdraws its influence over how the body partitions and stores energy.
One of the most clinically visible of these changes is a redistribution of adipose tissue. Under the influence of estrogen, fat is preferentially stored in subcutaneous depots, beneath the skin, particularly around the hips and thighs. As estrogen declines, that pattern shifts. Fat increasingly accumulates in visceral depots, deep within the abdominal cavity, surrounding the internal organs. As Dr. Judi Chervenak, M.D. describes it: the body often moves from a pear shape to an apple shape, with fat gathering around the midsection in ways that carry meaningfully different metabolic consequences than subcutaneous fat
Those consequences are significant. Visceral fat is not metabolically inert. It is an active endocrine tissue that secretes pro-inflammatory signaling molecules, including interleukin-6 (IL-6) and high-sensitivity C-reactive protein (hsCRP), into the circulation. The chronic low-grade inflammatory state that results, sometimes called inflammaging, creates a systemic environment that progressively impairs insulin sensitivity, disrupts lipid metabolism, and elevates cardiometabolic risk [4,5]. Simultaneously, estrogen withdrawal directly worsens insulin sensitivity through its own mechanisms, compounding the metabolic burden that visceral fat accumulation creates.
Together, these changes position menopause as a critical inflection point in the trajectory of metabolic health. The midlife acceleration in risk for type 2 diabetes, cardiovascular disease, and metabolic syndrome that epidemiological data consistently show in women is not coincidental to the timing of menopause. It is mechanistically connected to it [4,5].
A Coordinated Disruption, Not a Collection of Symptoms
What emerges from this picture is that menopause is not a collection of independent symptoms requiring separate management. It is a coordinated disruption in both hormonal signaling and cellular energy regulation, operating simultaneously across the brain, adipose tissue, vasculature, and metabolic systems. The symptoms that present clinically, cognitive changes, weight redistribution, insulin resistance, inflammatory burden, are downstream expressions of that upstream disruption.
This framing has direct therapeutic implications. HRT addresses the upstream hormonal withdrawal. GLP-1 receptor agonists address what that withdrawal leaves behind. As we will explore, the combination may offer something neither achieves alone.
GLP-1 Receptor Agonists: A New Framework for Metabolic Regulation
To understand why GLP-1 receptor agonists have generated such extraordinary clinical interest, it helps to start with the hormone they mimic. Glucagon-like peptide-1 is a signaling molecule released by specialized cells in the gut in response to food intake. Its natural role is to coordinate the body's response to a meal: stimulating insulin secretion, suppressing glucagon, slowing the rate at which the stomach empties its contents into the small intestine, and signaling to the brain that energy has arrived and appetite can be reduced. It is, in other words, a key node in the body's energy regulation network.
The problem with natural GLP-1 is that it is short-lived. Enzymes in the bloodstream degrade it within minutes of release, limiting its therapeutic potential. GLP-1 receptor agonists solve that problem by mimicking the hormone's structure while resisting rapid degradation, allowing sustained activation of GLP-1 receptors across multiple tissues for hours or days, depending on the formulation [6].
How They Work: Central and Peripheral Effects
The effects of GLP-1 receptor agonists operate on two levels simultaneously, and understanding both is important for appreciating why they are relevant to the menopausal metabolic transition described in the previous section.
At the central level, GLP-1 receptors are expressed in regions of the hypothalamus and brainstem that govern appetite and food-seeking behavior. Activating these receptors reduces what many patients describe as "food noise," the persistent, intrusive preoccupation with food and eating that makes sustained caloric restriction so difficult to maintain through willpower alone [7]. This is not simply appetite suppression in the conventional sense. It appears to represent a recalibration of the brain's reward and satiety circuitry, reducing the hedonic drive to eat independently of hunger signals.
At the peripheral level, GLP-1 receptor agonists slow gastric emptying, which blunts the rate at which glucose enters the bloodstream after meals and reduces postprandial glucose spikes. They improve insulin sensitivity in peripheral tissues, favorably shift lipid profiles, and exert anti-inflammatory effects across multiple organ systems. Collectively, these actions address several of the metabolic consequences that estrogen withdrawal sets in motion, including worsening insulin resistance, dyslipidemia, and chronic low-grade inflammation.
The Clinical Evidence
The scale of benefit demonstrated in clinical trials has been striking. In the STEP and SURMOUNT trial programs, participants treated with semaglutide and tirzepatide respectively achieved average weight reductions of 15 to 22% of total body weight, losses that had previously been achievable only through bariatric surgery [8,9]. Perhaps more significantly for long-term healthspan, these reductions were accompanied by meaningful improvements in cardiometabolic risk markers across the board.
The cardiovascular data are particularly compelling. In the SELECT trial, semaglutide reduced the incidence of major adverse cardiovascular events, including heart attack and stroke, by 20% in individuals with established cardiovascular disease and obesity but without diabetes [10]. This finding positioned GLP-1 receptor agonists not merely as metabolic drugs but as cardiovascular therapeutics, a distinction that carries particular relevance for menopausal women, whose cardiovascular risk accelerates significantly during and after the transition.
Where GLP-1s Fit in the Menopausal Framework
Returning to the framework established in the previous section, the menopausal transition represents a disruption operating on two levels: the upstream withdrawal of hormonal signaling, and the downstream metabolic consequences that withdrawal produces. GLP-1 receptor agonists operate at the downstream level, intervening in the metabolic environment that estrogen's absence creates. HRT operates upstream, restoring the hormonal signaling that menopause withdraws. They are complementary tools, not alternatives, and understanding why requires looking more closely at what HRT actually does at the mechanistic level.
Rethinking Hormone Replacement Therapy
GLP-1 receptor agonists, as the preceding section established, address the metabolic consequences of estrogen withdrawal with considerable potency. But they do not address the withdrawal itself. For that, the relevant therapy is the one that has been part of menopausal care for decades, and that has had a more complicated clinical history than almost any other intervention in women's medicine.
For most of the twentieth century, hormone replacement therapy occupied an uncomplicated position in women's medicine. Estrogen declined at menopause, symptoms followed, and restoring estrogen relieved them. The logic was straightforward, the clinical use widespread, and the benefits, at least for symptom management, well established.
Then, in 2002, a single study changed everything.
The Women's Health Initiative, a large randomized controlled trial, reported increased risks of breast cancer, cardiovascular disease, stroke, and thromboembolic events in women taking combined estrogen and progestin therapy [11]. The findings were published, the headlines followed, and HRT prescribing dropped dramatically almost overnight. For nearly two decades, the therapy that had been a cornerstone of menopausal care became something many clinicians and patients approached with significant caution or avoided entirely.
What subsequent analysis revealed, however, was that the WHI findings were far more context-dependent than the initial reporting suggested. The average age of participants in the original trial was 63, and many were more than a decade past menopause at the time of enrollment. This was not a population of women navigating the menopausal transition. It was a population of older postmenopausal women, a meaningfully different biological context, in whom initiating hormone therapy may carry different risks than initiating it earlier [11].
The Timing Hypothesis
This distinction gave rise to what is now called the timing hypothesis: the idea that the risks and benefits of HRT are critically dependent on when therapy begins relative to the onset of menopause [12]. Women who initiate HRT before age 60, or within approximately ten years of their final menstrual period, appear to have a substantially more favorable risk profile than those who begin later. In some analyses, early initiation is associated not merely with neutral cardiovascular outcomes but with active cardiovascular benefit, a finding that aligns with the known role of estrogen in maintaining vascular elasticity, endothelial function, and lipid metabolism [12].
The formulations themselves have also evolved considerably since the WHI era. Transdermal estradiol, delivered through patches or gels rather than oral tablets, bypasses the liver entirely, avoiding the first-pass hepatic metabolism that oral estrogen undergoes and that is associated with increased clotting factor production and thrombotic risk. Micronized progesterone, which more closely mirrors the molecular structure of the body's own progesterone, has largely replaced the older synthetic progestins used in the original WHI trial, and appears to offer improved tolerability and a more favorable safety profile [11,27]. The HRT of 2026 is, in meaningful ways, a different therapeutic proposition than the HRT evaluated in 2002.
The conventional framing of HRT as a symptom management tool, addressing hot flashes, sleep disruption, and genitourinary changes, captures only part of its biological relevance. Estrogen's role in the body extends well beyond reproductive function. It supports bone mineral density by regulating the balance between bone formation and resorption. It maintains vascular tone and endothelial health. It modulates lipid metabolism, influencing the balance between HDL and LDL cholesterol. And as established in the previous section, it plays an active role in neuronal energy metabolism and mitochondrial function.
When estrogen declines, each of these systems is affected. The visible symptoms of menopause, the hot flashes and the sleep disruption, are the acute manifestations of that withdrawal. The less visible consequences, accelerating bone loss, shifting lipid profiles, worsening insulin sensitivity, increasing visceral adiposity, and declining lean mass, are the slower-moving ones that accumulate over years and compound long-term health risk.
By restoring aspects of estrogen signaling, HRT does not merely suppress symptoms. It helps stabilize the broader physiological environment that estrogen had been maintaining, creating a more favorable metabolic context within which other interventions can operate. This is particularly relevant when considered alongside GLP-1 receptor agonists, which target the downstream metabolic consequences of estrogen withdrawal directly.
Which brings us to the central question we have been building toward. Menopause represents a coordinated disruption in both hormonal signaling and metabolic regulation. HRT addresses the former. GLP-1 receptor agonists address the latter.
Hormone Therapy and GLP-1 Medications: A Systems-Level Synergy
The case for combining HRT and GLP-1 receptor agonists does not rest simply on the observation that each addresses a different part of the menopausal disruption. It rests on something more specific: that these two therapies intersect on the same underlying biological pathways, and that their combined influence on those pathways may be meaningfully greater than either produces alone.
The Molecular Case for Combination: Shared Pathways, Amplified Effects
At the cellular level, estrogen and GLP-1 receptor agonists converge on two signaling pathways that sit at the center of metabolic regulation and cellular survival.
The first is the PI3K-Akt pathway, a molecular cascade that governs cell survival, growth, and the uptake of glucose in response to insulin. Think of it like a lock-and-key system at the cell's front door. Insulin arrives, fits the lock, and the door opens to let glucose in. Both estrogen and GLP-1 receptor activation stimulate this same pathway, but rather than simply unlocking the door, they help widen the doorway itself, making the cell more responsive to insulin's signal and better equipped to take up glucose efficiently. When both signals are present simultaneously, that doorway is wider and more reliably open than when either operates alone, promoting insulin sensitivity and protecting cells from the kind of metabolic stress that chronic energy dysregulation produces.
The second is AMPK, or AMP-activated protein kinase, which functions as the cell's master energy sensor. Think of it as a fuel gauge and emergency response system combined into one. When the gauge reads low, AMPK triggers a coordinated set of responses designed to restore balance: increasing glucose uptake, burning stored fat for fuel, stimulating the production of new mitochondria, and temporarily suspending energy-consuming processes that are not immediately essential. It is the cell's way of saying: energy is scarce, priorities have changed, adapt accordingly. Both estrogen and GLP-1 signaling engage this same system, helping to keep the fuel gauge calibrated and the emergency response primed. With age and hormonal withdrawal, that calibration drifts, and the cell becomes slower to recognize and respond to energy stress. Together, estrogen and GLP-1 receptor agonists help restore that sensitivity, maintaining the kind of metabolic flexibility that aging and hormonal withdrawal progressively erode [13,14].
This convergence extends beyond energy sensing into the domain of inflammation and oxidative stress. As established earlier, the menopausal transition is associated with a shift toward chronic low-grade inflammation, driven partly by the loss of estrogen's anti-inflammatory signaling and partly by the accumulation of metabolically active visceral fat. Both estrogen and GLP-1 receptor agonists independently reduce the production of pro-inflammatory cytokines and improve the balance between oxidative damage and antioxidant defense. Used together, their combined effect on inflammatory tone appears to be more comprehensive than either achieves independently, helping to limit the tissue damage and metabolic dysfunction that chronic inflammation drives over years and decades [13,14].
Importantly, each therapy addresses limitations of the other. Estrogen alone may not fully overcome insulin resistance in the setting of obesity, while GLP-1 therapies do not directly restore the hormonal environment that supports mitochondrial and tissue-level function. Together, they cover more of the biological terrain that menopause disrupts than either can alone. And of all the systems where that combined coverage matters most, one stands apart, both in terms of the stakes involved and the specificity of the biological mechanisms at play. The brain.
Neuroprotection and Brain Energy Metabolism
Of all the systems affected by the menopausal transition, the brain may be where the combined case for HRT and GLP-1 receptor agonists is most compelling. And it is also where the stakes are highest.
As established earlier, estrogen withdrawal creates a measurable energetic deficit in the brain, impairing the ability of neurons to efficiently utilize glucose and leaving them increasingly vulnerable to oxidative stress and metabolic dysfunction. That vulnerability, sustained over years, has been linked to the early biological processes associated with neurodegeneration. The question is whether the combination of estrogen restoration and GLP-1 receptor activation can meaningfully support the brain's resilience during this window.
The biology suggests it can, and through several distinct but complementary mechanisms.
A Coordinated Defense for Neuronal Energy
Both estrogen and GLP-1 receptors are expressed in the brain regions most critical to cognition, including the hippocampus, the structure central to memory formation, and the prefrontal cortex, which governs executive function and decision-making [2]. Their presence in these regions is not incidental. Both hormones actively regulate the energy metabolism of the neurons within them.
Estrogen enhances mitochondrial efficiency in neurons, strengthening the antioxidant defenses that protect mitochondrial DNA from oxidative damage and helping maintain the structural integrity of the mitochondrial network. GLP-1 receptor activation complements this by promoting mitochondrial turnover, the process by which damaged or dysfunctional mitochondria are cleared and replaced, and by reducing the production of reactive oxygen species, the unstable molecules that accumulate during metabolic stress and accelerate cellular damage.
Think of it this way: estrogen helps keep the brain's power plants running cleanly and efficiently, while GLP-1 signaling ensures that aging or damaged power plants are replaced before they become a liability. Together, they support the kind of stable, sustained neuronal energy production that is increasingly recognized as a critical factor in preventing the early metabolic dysfunction associated with neurodegenerative disease [2].
Quieting Neuroinflammation
Beyond energy metabolism, both pathways converge on neuroinflammation, the activation of the brain's resident immune cells, known as microglia, in response to stress, injury, or metabolic disruption. In the short term, microglial activation is a protective response. Sustained over years, however, chronic neuroinflammation becomes damaging in its own right, contributing to the accumulation of amyloid plaques and tau protein tangles that are the defining pathological features of Alzheimer's disease.
Both estrogen and GLP-1 receptor signaling independently reduce microglial activation and the production of pro-inflammatory cytokines within the brain. Used together, their combined effect on neuroinflammatory tone may be more comprehensive than either achieves alone, helping to slow the inflammatory processes that accelerate neurodegeneration during and after the menopausal transition.
Supporting Synaptic Longevity
The third mechanism operates at the level of the synapse, the junction between neurons across which learning, memory, and cognition depend. Estrogen plays a well-established role in maintaining synaptic structure and density, particularly in the hippocampus, where it supports the physical connections between neurons that encode and retrieve memories. As estrogen declines, synaptic integrity in these regions can deteriorate, contributing to the cognitive changes many women experience during and after menopause.
GLP-1 receptor activation engages a complementary set of molecular pathways that support synaptic function from a different angle. It enhances the activity of CREB, a transcription factor that regulates the expression of genes involved in memory consolidation, and promotes the production of BDNF, or brain-derived neurotrophic factor, a protein that acts as a kind of molecular fertilizer for neurons, supporting their survival, growth, and the formation of new synaptic connections [15]. Where estrogen helps maintain the existing architecture of synaptic networks, GLP-1 signaling supports the biological machinery that allows those networks to adapt, strengthen, and repair.
Together, these three mechanisms, coordinated neuronal energy support, reduced neuroinflammation, and complementary synaptic maintenance, make the combination of HRT and GLP-1 receptor agonists particularly relevant for the preservation of cognitive resilience during the menopausal transition and beyond. But the hormonal environment of menopause is more complex than an estrogen and GLP-1 equation alone. A complete clinical picture requires accounting for the other hormones whose decline shapes how both therapies perform in practice, and in some cases, how effectively they protect the very systems this section has been examining.
The Full Hormonal Picture: Progesterone, Testosterone, and the Limits of a Two-Variable Equation
The framework built so far has focused primarily on estrogen and GLP-1 receptor agonists as the central players in the menopausal metabolic disruption. But the hormonal environment of menopause is more complex than a two-variable equation, and a complete picture requires accounting for the other hormones whose decline shapes how both therapies perform in practice.
The Role of Progesterone
For women with an intact uterus, progesterone is not optional within an HRT regimen. Estrogen administered without progesterone stimulates the endometrial lining, increasing the risk of endometrial hyperplasia and cancer. Progesterone provides the necessary counterbalance, protecting the uterine lining while estrogen exerts its broader systemic effects.
But progesterone's relevance extends beyond endometrial protection. Micronized progesterone, the bioidentical form that more closely mirrors the body's own hormone, has been associated with improvements in sleep quality and mood stability, two dimensions of menopausal health that carry their own metabolic consequences. Sleep, in particular, is not a passive recovery state. It is an active regulatory period during which the body manages appetite hormones, clears metabolic waste from the brain, and restores insulin sensitivity. Disrupted sleep elevates ghrelin, the hormone that drives hunger, while suppressing leptin, the hormone that signals satiety. For women using GLP-1 receptor agonists, whose therapeutic benefit depends significantly on recalibrating appetite regulation, chronic sleep disruption can work directly against the therapy's mechanism. In this sense, progesterone's role in supporting sleep is not merely a quality-of-life consideration. It is a factor that may meaningfully influence how effectively GLP-1 therapies perform.
The Overlooked Role of Testosterone
Testosterone is rarely the first hormone that comes to mind in discussions of women's health, but its decline during the menopausal transition carries consequences that are directly relevant to the metabolic framework this article has been building.
In women, testosterone contributes to the maintenance of lean muscle mass, bone density, libido, and overall physical vitality. Its decline accelerates the sarcopenic process that estrogen withdrawal initiates, reducing the metabolically active tissue that both burns energy at rest and supports functional independence in later life. This becomes particularly relevant in the context of GLP-1 receptor agonist therapy, where the substantial weight loss these medications produce is not exclusively fat loss. The body composition implications of that trade-off, and what testosterone can do to address them, are examined in detail in the combination strategy section that follows.
Hormonal Context Is Not Peripheral
What these considerations collectively reveal is that hormonal context is not a secondary variable in how metabolic therapies perform. It is a central one. The effectiveness of GLP-1 receptor agonists in menopausal women is shaped by the broader hormonal environment within which they operate: whether estrogen has been restored, whether progesterone is supporting sleep quality and endometrial health, and whether testosterone decline is accelerating the muscle loss that weight reduction can compound.
A therapeutic approach that addresses GLP-1 targets without attending to that hormonal context is working with one hand tied behind its back. The mechanistic case for integration is compelling. The critical next question is whether these interactions translate into measurable clinical benefit in real-world populations, and what the emerging evidence has to say about that.
What human data say so far (and what they don’t): Early Clinical Signals of GLP-1 and Hormone Therapy Synergy
The biological rationale for combining HRT and GLP-1 receptor agonists is, as the preceding sections have established, mechanistically coherent. Shared signaling pathways, complementary effects on inflammation and oxidative stress, coordinated neuroprotection, and mutually reinforcing influences on body composition all point in the same direction. But mechanistic plausibility is not clinical proof. The question that ultimately matters is whether this convergence produces measurably better outcomes in real women.
The human data are early. But the signals are notable.
A First Look at the Numbers
A 2026 study published in The Lancet Obstetrics, Gynaecology, and Women's Health took a direct look at this question, evaluating 120 postmenopausal women treated with tirzepatide, one of the most effective GLP-1 and GIP dual receptor agonists currently available [16]. Participants were divided into those receiving concurrent hormone therapy and those who were not, with groups carefully matched by age, BMI, and clinical history to reduce the most obvious confounding variables.
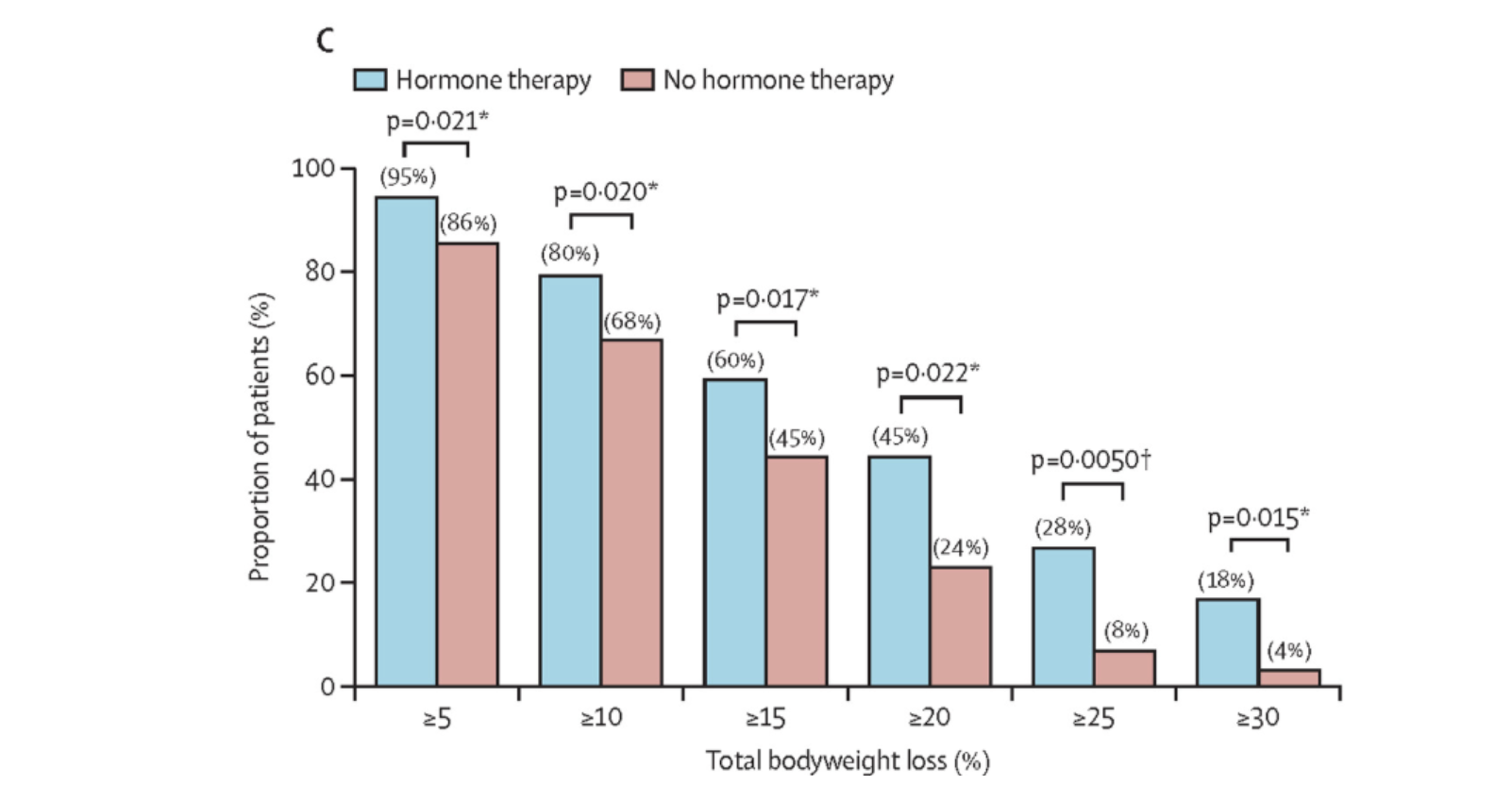
The results showed a consistent advantage for the combination, and that advantage became more pronounced at higher weight loss thresholds, precisely where GLP-1 therapy tends to be most clinically meaningful:
At a 20% weight loss threshold, 45% of women in the combination group achieved this level of loss, compared to 24% in the GLP-1 alone group. At 25%, the gap widened further: 28% versus 8%. At 30%, it widened again: 18% versus 4%.
These are not marginal differences. At the highest threshold, women receiving combination therapy were more than four times as likely to achieve 30% weight loss as those receiving tirzepatide alone. If replicated in larger prospective trials, that magnitude of difference would carry significant clinical implications.
It is important to be precise about what this study does and does not establish. As a retrospective analysis, it cannot prove causality. Women who were already receiving hormone therapy may have differed from those who were not in ways that the matching process did not fully capture: differences in baseline metabolic health, in how long they had been postmenopausal, in their physical activity levels, or in other clinical factors that influence GLP-1 responsiveness. These limitations are real, and they mean the findings should be interpreted as hypothesis-generating rather than definitive.
What the study does provide is a clinical signal that aligns with the mechanistic story. The pattern of results, particularly the amplification of benefit at higher weight loss thresholds, is consistent with the hypothesis that HRT restores a metabolic environment that makes GLP-1 therapy more effective: improving insulin sensitivity, preserving lean mass, reducing the inflammatory burden that blunts metabolic responsiveness, and supporting the mitochondrial function that underlies energy regulation. These are precisely the mechanisms that the preceding sections identified as potential synergy points.
The honest answer is that the field is at an early but genuinely promising juncture. The mechanistic case for combining HRT and GLP-1 receptor agonists in menopausal women is strong. The first human data, while preliminary, point in the direction the biology predicts. What is now needed are larger, prospective, randomized trials that can establish causality, characterize which women benefit most, identify the optimal timing and formulation of each therapy, and track outcomes across the full range of health domains this article has examined, metabolic, cardiovascular, cognitive, and functional.
That evidence is still being generated. But the conversation has already shifted. The question is no longer whether combining these therapies is biologically plausible. It is how to do so most effectively, for whom, and when.
At the highest threshold, women receiving combination therapy were more than four times as likely to achieve 30% weight loss as those receiving tirzepatide alone.
Personalizing Hormone Replacement Therapy and GLP-1 Combinations
The Lancet data make the case for combination therapy in quantitative terms. Women on hormone therapy were more than four times as likely to achieve 30% weight loss with tirzepatide than those who were not. But a statistic is not a clinical strategy. Translating that signal into real-world practice requires moving beyond the question of whether to combine these therapies toward the more demanding question of how, for which patients, at which stage of the transition, and with which specific formulations.
The goal of this integrated approach is not simply symptom relief or weight loss in isolation. It is the targeted modulation of the biological systems that drive metabolic decline, body composition changes, and long-term healthspan during and after the menopausal transition. That is a more ambitious objective than either therapy was originally designed to meet, and achieving it requires moving beyond one-size-fits-all prescribing toward a more individualized framework.
Several principles guide that framework.
Integration Produces Measurably Greater Outcomes
The first principle is that combining these therapies appears to produce results that neither achieves alone. Early clinical data in postmenopausal populations suggest that HRT combined with GLP-1 receptor agonist therapy is associated with total body weight loss approaching approximately 19%, compared to roughly 14% with GLP-1 therapy alone [16]. Additional emerging data further support the idea that hormonal status influences both weight loss responsiveness and body composition outcomes during GLP-1 therapy [17], consistent with the mechanistic picture of HRT restoring a metabolic environment that makes GLP-1 receptor agonists more effective.
The implication is that clinicians managing menopausal women on GLP-1 therapies should consider hormonal status not as a separate clinical question but as a variable that directly shapes metabolic treatment outcomes.
Formulation and Route of Delivery Are Not Interchangeable
The second principle is that how hormones are delivered matters as much as whether they are delivered. Not all HRT formulations carry the same risk profile, and those differences are clinically significant in the context of the cardiometabolic goals this combination approach is designed to serve.
Transdermal estradiol, delivered through patches or gels, bypasses hepatic first-pass metabolism and is associated with meaningfully lower thrombotic risk than oral estrogen formulations [18]. For women whose GLP-1 therapy is partly motivated by cardiovascular risk reduction, this distinction is not trivial. Similarly, micronized progesterone demonstrates a more favorable cardiometabolic and neurocognitive profile than the synthetic progestins used in older HRT formulations [19], making it the preferred choice in a framework explicitly aimed at supporting brain health and metabolic resilience alongside symptom management.
Formulation decisions are therefore not merely pharmacological details. They are strategic choices that either align with or work against the broader healthspan objectives of the combined approach.
Different Hormonal Strategies Target Different Physiological Domains
The third principle is that the specific combination of hormones prescribed shapes which aspects of the menopausal disruption are most directly addressed. Estrogen primarily targets mitochondrial function, vascular health, neuronal energy metabolism, and the redistribution of adipose tissue. Progesterone contributes endometrial protection, sleep quality, and mood stability. Testosterone, where indicated, supports lean mass preservation and physical resilience, particularly relevant during the weight loss that GLP-1 therapy produces. The clinical permutation chosen should therefore reflect not just a woman's symptom profile but her metabolic risk factors, body composition goals, cognitive health priorities, and cardiovascular history.
Monitoring Is Not Optional
The fourth principle is that this level of biological ambition requires a correspondingly rigorous approach to monitoring. Combining therapies that each exert significant effects on metabolic regulation, inflammation, and body composition creates a dynamic clinical picture that cannot be adequately managed through periodic symptom check-ins alone. Biomarkers including HbA1c, fasting insulin, lipid profiles, and body composition measurements provide the quantitative feedback needed to determine whether the combined approach is producing the intended metabolic effects, whether adjustments to formulation or dosing are needed, and whether the balance between the two therapies remains appropriate as a woman's physiology evolves through and beyond the menopausal transition.
Together, these four principles shift the clinical conversation from a question of which therapy to use toward a more sophisticated question of how to combine them most effectively, for which patient, at which stage of the transition, and toward which specific physiological objectives. In practice, that translates into three distinct clinical permutations, each shaped by a woman's hormonal status, surgical history, and metabolic profile. They are not interchangeable. Each addresses a different configuration of the menopausal disruption, and understanding what distinguishes them is essential to applying a complete integrated framework.
A. Estrogen-Only Therapy Combined with GLP-1 Receptor Agonists
For women who have undergone hysterectomy, the hormonal equation simplifies in one important respect: without a uterus, progesterone is not required for endometrial protection, making estrogen-only therapy the appropriate hormonal foundation. In this context, the combination of estrogen therapy and GLP-1 receptor agonists represents perhaps the most metabolically direct version of the integrated approach this article has been building toward.
Why the Combination Works
Estrogen's metabolic roles, established in earlier sections, are directly relevant here. It enhances insulin sensitivity in peripheral tissues, supports mitochondrial efficiency in both muscle and brain, and actively regulates how the body partitions fat between subcutaneous and visceral depots. Its decline during menopause is strongly and mechanistically associated with the shift toward central adiposity and the progressive metabolic inflexibility that follows [20]. Restoring estrogen signaling through therapy does not merely slow that shift. It addresses one of its primary biological drivers.
GLP-1 receptor agonists complement this from the downstream direction. Where estrogen works to restore the hormonal environment that governs fat distribution and metabolic regulation, GLP-1 therapy directly reduces caloric intake by recalibrating appetite signaling, improves glycemic control through multiple mechanisms, and drives the reduction of visceral fat mass that estrogen alone cannot reliably produce in the setting of significant metabolic burden.
The clinical data support this complementarity. In postmenopausal women, estrogen therapy has been associated with reductions in central adiposity and improvements in glucose metabolism independent of weight loss. GLP-1 receptor agonists, as established in earlier sections, independently produce average weight reductions of 15 to 22% of total body weight in the STEP and SURMOUNT trial programs [8,9]. Emerging evidence suggests that women receiving hormone therapy demonstrate more favorable weight loss trajectories and body composition outcomes when treated with GLP-1 based therapies than those who are not, particularly in women with preserved metabolic flexibility [16,17]. The combination appears to create a physiological environment in which each therapy performs better than it would alone.
Clinical considerations: Transdermal delivery is often preferred to reduce thrombotic risk. Individual risk factors, including migraine with aura or hormone-sensitive cancer history, should guide decision-making.
B. Estrogen + Progesterone + GLP-1
For women with an intact uterus, the hormonal framework expands necessarily to include progesterone alongside estrogen. But progesterone's role in this combination is not merely protective. It is, in its own right, a biologically active contributor to the metabolic and neurological goals that this integrated approach is designed to serve.
Why the Combination Works
Estrogen's contributions to this framework have been established throughout this article: metabolic regulation, mitochondrial support, vascular health, fat distribution, and neuronal energy metabolism. In women with an intact uterus, progesterone provides the essential endometrial counterbalance that makes sustained estrogen therapy safe. But micronized progesterone, the bioidentical form now preferred in modern HRT protocols, does considerably more than protect the uterine lining.
One of its most clinically relevant effects in the context of GLP-1 therapy is its influence on sleep. Micronized progesterone has been associated with improved sleep architecture and reductions in nighttime awakenings [21], an effect that appears to involve its interaction with GABA receptors in the brain, the same receptor system targeted by many sedative medications, but through a gentler and more physiologically integrated mechanism. This matters more than it might initially appear.
Sleep, in particular, is not a passive recovery state. It is an active regulatory period during which the body manages appetite hormones and restores insulin sensitivity. Progesterone's role in supporting sleep quality is therefore not merely a quality-of-life consideration. As explored in the combination strategy section that follows, it may meaningfully influence how effectively GLP-1 therapies perform. Progesterone's sleep-supporting effects therefore have downstream metabolic consequences that may meaningfully enhance GLP-1 responsiveness in ways that are easy to underestimate.
Beyond sleep, combined estrogen and progesterone therapy has been linked to improvements in insulin sensitivity and lipid profiles in women during early menopause [22], adding a further layer of metabolic benefit to the combination. And emerging data suggest that optimizing the hormonal environment during the menopausal transition more broadly may positively influence weight loss trajectories and body composition outcomes during GLP-1 treatment [17], consistent with the mechanistic picture of restored hormonal signaling creating a more metabolically receptive physiological state.
Clinical considerations: Formulation matters. Micronized progesterone is generally preferred over synthetic progestins due to a more favorable cardiovascular and neurocognitive profile. Transdermal estrogen remains the preferred route in many cases.
C. Testosterone + GLP-1 (Selected Cases)
Among the hormonal considerations in menopausal metabolic care, testosterone is the most frequently overlooked. It is rarely the first hormone discussed in the context of women's health, and its decline during the menopausal transition receives far less clinical attention than the fall in estrogen and progesterone. But in the specific context of GLP-1 receptor agonist therapy, testosterone's role becomes particularly consequential, for a reason that goes to the heart of what these medications are actually trying to achieve.
The Body Composition Problem with Rapid Weight Loss
GLP-1 receptor agonists are remarkably effective at producing weight loss. That is, by now, well established. What is less commonly discussed is the composition of that weight loss. When the body loses mass rapidly, it does not draw exclusively from fat stores. Studies suggest that up to 25 to 40% of the weight lost during GLP-1 therapy may come from lean mass, primarily skeletal muscle, if active steps are not taken to preserve it [23]. For a menopausal woman already navigating the muscle loss that estrogen withdrawal and the aging process impose, that trade-off carries significant long-term consequences.
Skeletal muscle is not simply the tissue that moves the body. It is the primary site of insulin-mediated glucose disposal, meaning that losing it worsens the very insulin resistance that GLP-1 therapy is designed to improve. It is metabolically active at rest, contributing meaningfully to basal energy expenditure. And it is the biological substrate of physical strength, balance, and functional independence, the capacities that determine quality of life in the later decades. Losing a substantial fraction of it in the process of achieving weight loss is, in metabolic terms, trading one problem for another.
Where Testosterone Fits
Testosterone contributes directly to the maintenance of lean muscle mass, bone density, neuromuscular function, and physical vitality. Its decline during menopause accelerates the sarcopenic trajectory that estrogen withdrawal initiates, and its restoration, where clinically appropriate, may help counteract the muscle loss that rapid weight loss compounds.
The 2019 Global Consensus Position Statement on testosterone use in women established that testosterone therapy is associated with improvements in lean mass, bone density, and sexual function in postmenopausal women [24]. While direct combination data with GLP-1 receptor agonists remain limited and still evolving, the mechanistic rationale is straightforward: if GLP-1 therapy is driving significant weight loss and a meaningful fraction of that loss would otherwise come from muscle, then supporting testosterone levels provides a biologically coherent way to shift the composition of that loss toward fat rather than lean tissue.
Think of it this way: GLP-1 therapy opens the door to significant fat reduction, but without attention to muscle preservation, some of what walks out that door is tissue the body cannot easily afford to lose. Testosterone helps keep that tissue in place while the fat reduction proceeds.
Clinical considerations: Therapy should be carefully monitored due to dose sensitivity and potential androgenic effects. This approach is best suited for women with clear clinical indications, such as low libido, reduced strength, or significant lean mass loss.
Cognitive Health and the Prevention of Alzheimer’s Disease
The conversation around HRT and GLP-1 receptor agonists has so far focused primarily on what is most immediately measurable: weight loss, body composition, insulin sensitivity, and cardiovascular risk. These are important outcomes, and the evidence supporting the combination in these domains is growing. But the biology established throughout this article points toward a consequence of the menopausal transition that may ultimately prove more significant than any of them.
Alzheimer's disease affects women at roughly twice the rate of men. It is the leading cause of dementia globally, and despite decades of research, it remains without a disease-modifying treatment. The hormonal biology of menopause is increasingly understood as a contributing factor to that disparity, and the convergence of estrogen and GLP-1 signaling in the brain offers a mechanistic framework for understanding why, and potentially what to do about it.
The conventional understanding of Alzheimer's disease frames it primarily as a problem of protein accumulation: amyloid plaques building up between neurons, tau tangles forming within them, and the resulting structural damage driving cognitive decline. That framing is not wrong, but it is increasingly understood to be incomplete. A growing body of evidence positions Alzheimer's disease as fundamentally a disorder of impaired brain energy metabolism, one in which neurons progressively lose the ability to utilize glucose efficiently, creating an energetic crisis that precedes structural damage by years or even decades.
This reframing has led some researchers to describe Alzheimer's as a form of brain insulin resistance, sometimes referred to informally as Type 3 Diabetes. The terminology is contested, but the underlying observation is not: the metabolic deterioration of neurons, their diminishing capacity to produce and manage energy, appears to be an early and potentially modifiable feature of the disease process, not merely a downstream consequence of it.
This is where the convergence of estrogen and GLP-1 signaling becomes particularly relevant to cognitive aging.
Two Systems, One Target
As established in earlier sections, both estrogen and GLP-1 receptors are expressed in the brain regions most critical to cognition and memory. Both influence mitochondrial function, inflammatory tone, and the molecular machinery that governs neuronal survival and repair, engaging the same PI3K-Akt signaling pathway established earlier, now operating within neurons rather than peripheral tissue. What emerges is a coherent biological framework: estrogen maintains the mitochondrial infrastructure and synaptic stability that neurons depend on for sustained function, while GLP-1 receptor activation reduces the inflammatory burden that progressively degrades that infrastructure. Together, they address both the energy supply side and the inflammatory stress side of the neuronal resilience equation simultaneously.
Targeted Neuroprotection: GLP-1 and Estrogen Conjugates
Emerging research is beginning to exploit this convergence in a more direct and targeted way. Lizcano and colleagues investigated a novel class of molecules called GLP-1 and estrogen conjugates, engineered compounds that chemically link GLP-1 and estrogen into a single therapeutic agent [2]. The rationale is elegant: because GLP-1 receptors are enriched in metabolically active brain regions, using GLP-1 as a delivery vehicle allows estrogen to be directed preferentially toward the central nervous system, potentially enhancing its neuroprotective effects while limiting systemic hormonal exposure.
Complementary preclinical work by Sanchez-Garrido and colleagues evaluated these conjugates in models of metabolic and hormonal dysfunction, identifying molecular changes across pathways directly implicated in neurodegeneration [25]. The findings paint a consistent picture of cellular remodeling in a protective direction.
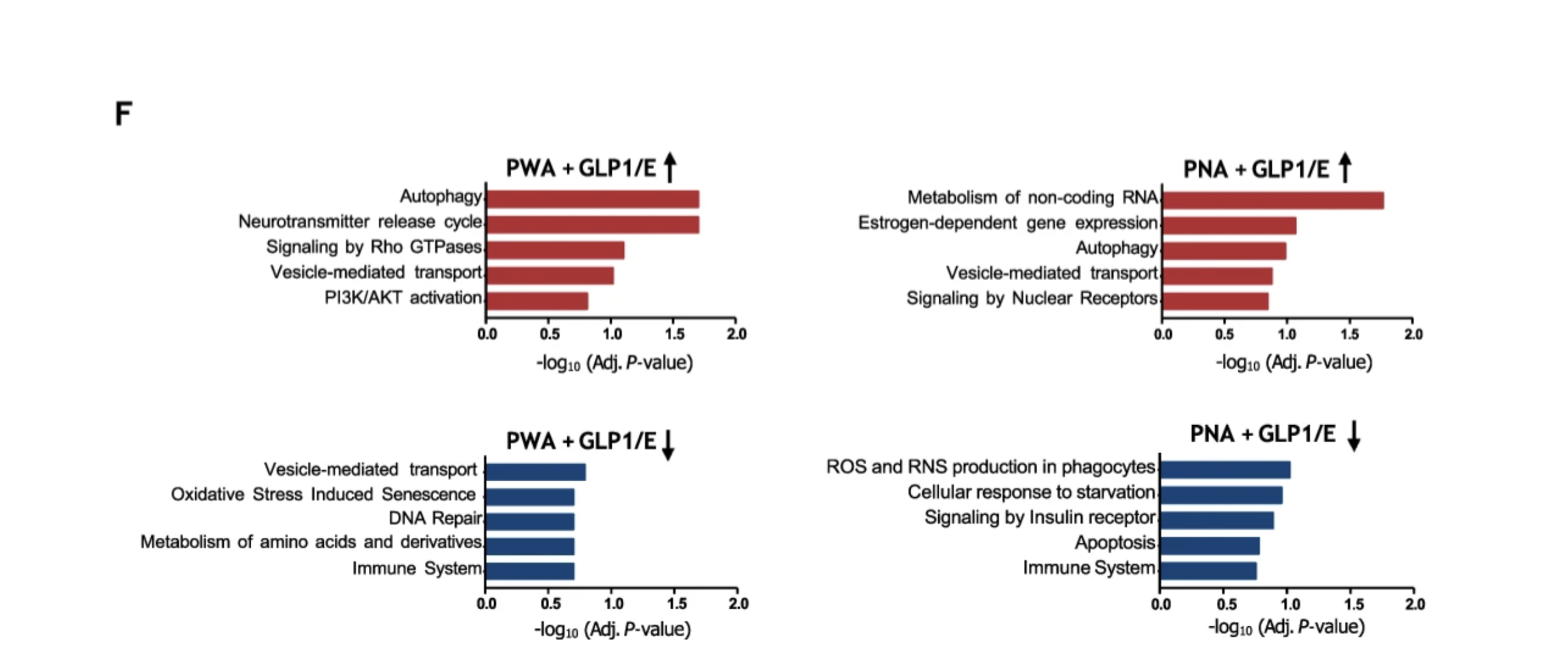
Bar graph shows which biological processes the brain is doing more or less after treatment. The red bars pointing to the right represent upregulated pathways, meaning the treatment made these processes more active. These include vital health functions like autophagy and cell signaling (‘PI3K/AKT activation’). The blue bars at the bottom represent downregulated pathways, meaning the treatment slowed these processes down. These include harmful processes like oxidative stress (‘Oxidative stress induced senescence’) and cell death (‘Apoptosis’). The length of the bar shows how strong the statistical evidence was for that change.
Among the key observed effects, several stand out as particularly relevant to the Alzheimer's disease framework. Enhanced autophagy, the cellular process by which damaged or misfolded proteins are identified and cleared, was observed, suggesting improved capacity to remove the amyloid and tau aggregates that accumulate in neurodegeneration. Activation of PI3K-Akt signaling supported neuronal survival and repair through the pathway established earlier in this article. Improvements in neurotransmitter cycling supported synaptic function and memory consolidation. And reductions in reactive oxygen and nitrogen species pointed to a meaningful decrease in neuroinflammatory burden, one of the central drivers of progressive neuronal injury in Alzheimer's disease.
Taken together, these findings suggest that combining GLP-1 and estrogen signaling does not simply add two neuroprotective effects together. It may activate a more integrated cellular response, one that simultaneously improves energy metabolism, clears pathological protein accumulations, reduces inflammation, and supports the synaptic architecture that cognition depends on.
A Converging Model of Brain Resilience
Building on these mechanisms, a unifying model begins to emerge: estrogen and GLP-1 signaling converge on shared processes that govern neuronal energy balance, inflammatory tone, and cellular survival.
Within this framework:
- HRT supports neural bioenergetics and mitochondrial stability, helping sustain adequate energy supply
- GLP-1 therapies reduce systemic and central inflammatory stress, improving the environment in which neurons function
Rather than acting in isolation, these therapies may operate synergistically to stabilize both energy production and stress response systems within the brain. This integrated effect provides a biologically plausible pathway for reducing vulnerability to cognitive decline and neurodegeneration over time.
A Note on Where the Science Stands
It is important to be clear about the current state of this evidence. The conjugate research and much of the mechanistic work described here remains preclinical, conducted in animal models and cell systems rather than in human clinical trials. The findings are compelling as proof of concept, and they are mechanistically coherent with what the human observational and clinical data suggest. But translating preclinical findings into established human therapeutic strategies requires a level of evidence that does not yet exist for the most novel aspects of this work.
What the preclinical data offer is a biologically grounded rationale for the direction of travel, and a framework for designing the human studies that will ultimately determine how much of this neuroprotective potential translates into clinical benefit. In the meantime, the combination of HRT and GLP-1 receptor agonists represents a strategy with a strong mechanistic case and growing clinical support across the metabolic and cardiovascular domains examined in earlier sections, with emerging evidence that the cognitive domain may prove equally important.
Translating any of this into practice, however, requires careful attention to safety, timing, and individual variability. It is to those practical clinical considerations that we turn next.
Clinical Risks and Considerations of Hormone Replacement Therapy
The case for combining HRT and GLP-1 receptor agonists is, as the preceding sections have established, biologically compelling. But the strength of a mechanistic argument does not override the need for careful clinical judgment, and both therapeutic approaches carry risk profiles that must be understood and managed individually. Translating the integrated framework this article has described into responsible clinical practice requires the same precision that the science demands.
HRT is widely used to address symptoms such as vasomotor instability, sleep disruption, and bone loss. However, its safety profile is closely tied to timing, formulation, and individual risk factors.
The timing hypothesis remains a central principle. Initiating estrogen therapy earlier in the menopausal transition, typically within 10 years of the final menstrual period and before age 60, is associated with a more favorable cardiovascular profile [26]. In contrast, starting therapy later, when vascular changes such as plaque accumulation are more advanced, may increase the risk of thrombotic events.
Mode of delivery also plays an important role. Oral estrogen undergoes first-pass metabolism in the liver, which can increase clotting factors and elevate the risk of venous thromboembolism. Transdermal estrogen bypasses this pathway and is therefore associated with lower thrombotic risk, making it a preferred option for many women, particularly those with elevated baseline risk [27].
Cancer risk must also be considered within the appropriate clinical context. Estrogen alone carries a different risk profile than combined estrogen-progesterone therapy. In women with an intact uterus, progesterone is required to prevent endometrial hyperplasia and reduce cancer risk [28]. Modern formulations and dosing strategies have improved safety, and current guidelines emphasize individualized decision-making rather than generalized avoidance.
GLP-1 Therapies: Safety and Clinical Considerations
GLP-1–based therapies, including newer dual- and triple-agonists targeting GLP-1, GIP, and glucagon pathways, offer substantial metabolic benefits but require thoughtful use [29].
Gastrointestinal effects such as nausea, early satiety, and changes in bowel habits are common, though typically transient and improved with gradual dose titration [30]. More clinically relevant, however, are the downstream effects of rapid weight loss and altered metabolic signaling.
As body weight decreases, the risk of gallstone formation increases, particularly with rapid fat loss. In addition, a proportion of weight loss often comes from lean mass, with implications for metabolic health, strength, and long-term function. Preserving muscle through adequate protein intake and resistance training is therefore essential, particularly in midlife populations already vulnerable to sarcopenia [31].
Rare but more serious risks, including pancreatitis, should be discussed. Because GLP-1 therapies delay gastric emptying, temporary discontinuation is recommended prior to procedures requiring anesthesia to reduce aspiration risk [32]. These therapies are also contraindicated during pregnancy due to potential effects on fetal development.
Combination Therapy: Safety and Clinical Considerations
When HRT and GLP-1 therapies are used together, the primary challenge shifts from isolated risk management to integrated physiologic balance. The combination can significantly reduce appetite and caloric intake, which, if not carefully managed, may lead to dehydration, fatigue, or low blood pressure particularly in individuals also taking antihypertensive medications. Ensuring adequate nutrition, especially sufficient protein intake, becomes critical to preserving lean mass and maintaining metabolic stability.
This combined approach also increases the need for consistent clinical monitoring. Blood pressure, glycemic markers, lipid profiles, and liver function should be evaluated regularly, alongside changes in body composition and symptom patterns. Subtle changes such as persistent fatigue, abnormal bleeding, or gastrointestinal symptoms can provide important early signals that adjustments may be needed.
Beyond Symptom Relief
From an ethical perspective, it is important to distinguish between therapeutic intent and superficial optimization. These interventions are not cosmetic tools, but targeted strategies designed to modulate underlying physiologic systems.
When used appropriately, the combination of HRT and GLP-1 therapies can meaningfully improve metabolic health, reduce cardiovascular risk, and support quality of life. However, these benefits depend on careful patient selection, informed consent, and ongoing clinical oversight.
The goal is not simply to reduce weight or alleviate symptoms, but to restore a more stable physiological state, one that supports resilience, function, and long-term healthspan.
Advancing from Weight Loss to Healthspan Outcomes
Establishing that a combined approach is safe and clinically meaningful is necessary, but it is not sufficient. The risk framework just outlined tells us what to monitor and what to avoid. What it cannot yet tell us is whether managing those risks successfully translates into the broader healthspan outcomes this article has been building toward: preserved cognitive function, durable cardiovascular resilience, sustained lean mass, and reduced long-term disease burden. That question requires a different kind of evidence, and it is largely still being generated.
While the combination of hormone replacement therapy and GLP-1 based medications shows strong early promise, much of the current evidence focuses on short-term weight outcomes rather than long-term effects on cardiovascular health, cognitive function, and overall healthspan
Future research will need to be more precise. Trials should stratify participants by hormone formulation, route of delivery, baseline metabolic health, and time since menopause. These variables meaningfully influence both efficacy and safety, including cardiovascular risk, thrombotic events, and metabolic response.
Equally important, outcome measures must evolve beyond body weight alone. Body composition analysis, including fat mass versus lean mass, should be standard. Longitudinal cardiometabolic panels can track glucose regulation, lipid dynamics, inflammation, and liver function. Brain health should also be incorporated, using cognitive testing and imaging to assess whether these therapies influence neurodegenerative risk. This is particularly relevant given the intersection of estrogen signaling, GLP-1 pathways, and genetic risk factors related to APOE4 status.
Emerging Therapies and Expanding Potential
The research agenda just outlined assumes a relatively stable therapeutic landscape, one in which the primary task is generating better evidence for the combinations already in use. But the landscape itself is not standing still. Even as the clinical community works to establish the long-term benefits of current GLP-1 and HRT combinations, a new generation of metabolic therapies is already moving through trials, ones that extend the biological logic of this article's central argument into territory that semaglutide and tirzepatide alone cannot reach.
Next-generation agents, including triple agonists such as retatrutide, extend beyond GLP-1 and GIP to include glucagon receptor activation, introducing a new dimension of energy expenditure and metabolic regulation.
In large clinical trials such as TRIUMPH-4, these agents have demonstrated substantial weight loss and improvements in inflammatory and cardiometabolic markers [33]. The results were provocative, weight loss with participants losing up to 28.7% of their total body weight over 68 weeks [33]. This therapy also showed massive improvements in comorbidities like knee pain and inflammatory cardiovascular markers such as high-sensitivity C-reactive protein. However, increased potency requires vigilance. These findings reinforce an important principle: greater pharmacologic strength does not always translate to optimal individual outcomes. As therapies become more powerful, personalization and long-term safety monitoring become even more critical. This highlights that the maximum dose is not always the optimal dose for every individual and underscores the need for longer follow-up to ensure the durability of these benefits.
Conclusion: Toward Integrated Healthspan Medicine
The pace of therapeutic innovation in this space is accelerating. But the most important shift may not be pharmacological. It may be conceptual.
Menopause has long been framed as a collection of symptoms to be managed individually, hot flashes addressed here, bone loss treated there, metabolic changes handled separately. What the biology reviewed in this article makes clear is that this framing has always been inadequate. Menopause is a coordinated disruption, one that withdraws hormonal signals that had been quietly governing cellular energy regulation, inflammatory tone, mitochondrial function, and tissue maintenance across virtually every system in the body simultaneously. Treating it as a series of isolated problems produces, at best, partial solutions.
The convergence of hormone replacement therapy and GLP-1 receptor agonists represents a meaningful departure from that approach. Rather than targeting symptoms in isolation, this combination addresses the full architecture of the menopausal disruption: the hormonal withdrawal and the metabolic environment it creates, simultaneously and through mechanisms that reinforce one another. Their shared activation of overlapping cellular pathways means the combined effect is not simply additive. It may be genuinely synergistic.
The early clinical data, while preliminary, point in the direction the biology predicts. And the domains of potential benefit extend well beyond what most people associate with either therapy. Metabolic health, cardiovascular resilience, cognitive function, and long-term protection against neurodegeneration are all plausibly within reach of a therapeutic strategy that addresses both the hormonal and metabolic dimensions of the menopausal transition simultaneously.
What is now needed is the evidence infrastructure to match the biological ambition. Larger prospective trials, stratified by hormone formulation, route of delivery, timing of initiation, and baseline metabolic health, will be required to establish causality, characterize which women benefit most, and track outcomes across the full range of health domains this article has examined. The science is moving in that direction. The clinical conversation has already shifted.
The goal was never simply to reduce weight or suppress symptoms. It is to restore a more stable physiological state, one that supports resilience, function, and the kind of long-term healthspan that the biology of menopause, left unaddressed, progressively erodes. That is a goal worthy of the integrated approach the evidence is beginning to support.
- Mukherjee, A., Ghaem-Maghami, S., & Syed, A. A. (2025). Use of incretin-based therapies in women using hormone replacement therapy (HRT). Post reproductive health, 31(2), 127–131. https://doi.org/10.1177/20533691251343069
- Lizcano F, Sanabria D and Aviles E (2025) Hormonal modulation, mitochondria and Alzheimer’s prevention: the role of GLP-1 agonists and estrogens. Front. Mol. Biosci. 12:1622186. doi: 10.3389/fmolb.2025.1622186 https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2025.1622186/full
- Leri M, Bertolini A, Diaz M and Marongiu R (2025) Editorial: Estrogens and neurodegeneration: a link between menopause and Alzheimer’s disease in women. Front. Mol. Biosci. 12:1727385. doi: 10.3389/fmolb.2025.1727385 https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2025.1727385/full
- Palacios S. (2025). Implications of the era of incretin-based weight loss therapy in menopause. Expert review of endocrinology & metabolism, 20(6), 441–443. https://doi.org/10.1080/17446651.2025.2540000
- Graczyk, N. A., & Bisschops, J. (2026). Glucagon-Like Peptide-1 Receptor Agonists (GLP-1RAs) for Obesity and Symptoms in Menopause: A Review. Cureus, 18(1), e101693. https://doi.org/10.7759/cureus.101693
- Graczyk, N. A., & Bisschops, J. (2026). Glucagon-like peptide-1 receptor agonists (GLP1s) for obesity and symptoms in menopause: A review. Cureus, 18(1), e101693. https://doi.org/10.7759/cureus.101693
- Kim, K. S., Park, J. S., & Choi, H. J. (2025). Mechanisms of glucagon-like-peptide 1 in the brain beyond metabolic effects. Annals of pediatric endocrinology & metabolism, 30(4), 165–174. https://doi.org/10.6065/apem.2448320.160
- Wilding, J. P. H., Batterham, R. L., Calanna, S., Davies, M., Van Gaal, L. F., Lingvay, I., McGowan, B. M., Rosenstock, J., Tran, M. T. D., Wadden, T. A., Wharton, S., Yokote, K., Zeuthen, N., & Kushner, R. F. (2021). Once-weekly semaglutide in adults with overweight or obesity. The New England Journal of Medicine, 384(11), 989–1002. https://doi.org/10.1056/NEJMoa2032183
- Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., Wharton, S., Connery, L., Alves, B., Kiyosue, A., Zhang, S., Liu, B., Bunck, M. C., Stefanski, A., & Tirzepatide Once Weekly for the Treatment of Obesity Investigators. (2022). Tirzepatide once weekly for the treatment of obesity. The New England Journal of Medicine, 387(3), 205–216. https://doi.org/10.1056/NEJMoa2206038
- Lincoff, A. M., Brown-Frandsen, K., Colhoun, H. M., Deanfield, J., Emerson, S. S., Frias, J. P., Leiter, L. A., Lingvay, I., Marso, S. P., Rosenstock, J., Wilding, J. P. H., & SELECT Trial Investigators. (2023). Semaglutide and cardiovascular outcomes in obesity without diabetes. The New England Journal of Medicine. Advance online publication. https://doi.org/10.1056/NEJMoa2307563
- Cagnacci, A., & Venier, M. (2019). The Controversial History of Hormone Replacement Therapy. Medicina (Kaunas, Lithuania), 55(9), 602. https://doi.org/10.3390/medicina55090602
- Mehta, J. M., Chester, R. C., & Kling, J. M. (2019). The Timing Hypothesis: Hormone Therapy for Treating Symptomatic Women During Menopause and Its Relationship to Cardiovascular Disease. Journal of women's health (2002), 28(5), 705–711. https://doi.org/10.1089/jwh.2018.7201
- Lucente, D., Bellino, S., & La Salvia, A. (2025). GLP-1 Receptor Agonists in Solid Tumour Therapy: Exploring Their Anticancer Potential and Underlying Molecular Pathways. Genes, 16(11), 1352. https://doi.org/10.3390/genes16111352
- Kazi, A. A., Molitoris, K. H., & Koos, R. D. (2009). Estrogen rapidly activates the PI3K/AKT pathway and hypoxia-inducible factor 1 and induces vascular endothelial growth factor A expression in luminal epithelial cells of the rat uterus. Biology of reproduction, 81(2), 378–387. https://doi.org/10.1095/biolreprod.109.076117
- Richard, J. E., Mohammad, A., Go, K. A., McGovern, A. J., Rechlin, R. K., Splinter, T. F. L., Lieblich, S. E., Radovic, L. K., Feng, L., Blankers, S. A., Yang, B., Douros, J. D., Finan, B., & Galea, L. A. M. (2025). Sex-specific metabolic and central effects of GLP-1–estradiol conjugate in middle-aged rats on a standard or western diet. Brain Behavior and Immunity, 106088–106088. https://doi.org/10.1016/j.bbi.2025.106088
- Castaneda, R., Bechenati, D., Tama, E., Rivera Gutierrez, R., Espinosa, M. A., Villamarin, J., Rajjo, T. I., Acosta, A., Faubion, S., Shufelt, C., & Hurtado Andrade, M. D. (2026). The role of menopause hormone therapy in modulating tirzepatide-associated weight loss in postmenopausal women with overweight or obesity: A retrospective cohort study. The Lancet Obstetrics, Gynaecology & Women’s Health, 2(2), e118–e128. https://doi.org/10.1016/S3050-5038(25)00145-1
- Younglove, C. (2026). Clinical Review: Menopause Hormone Therapy in Weight Management. Obesity Pillars, 100258.
- Canonico, M., Oger, E., Plu-Bureau, G., Conard, J., Meyer, G., Lévesque, H., Trillot, N., Barrellier, M. T., Wahl, D., Emmerich, J., Scarabin, P. Y., & Estrogen and Thromboembolism Risk (ESTHER) Study Group (2007). Hormone therapy and venous thromboembolism among postmenopausal women: impact of the route of estrogen administration and progestogens: the ESTHER study. Circulation, 115(7), 840–845. https://doi.org/10.1161/CIRCULATIONAHA.106.642280
- Stute, P., Wildt, L., & Neulen, J. (2018). The impact of micronized progesterone on breast cancer risk: a systematic review. Climacteric : the journal of the International Menopause Society, 21(2), 111–122. https://doi.org/10.1080/13697137.2017.1421925
- Mauvais-Jarvis, F., Clegg, D. J., & Hevener, A. L. (2013). The role of estrogens in control of energy balance and glucose homeostasis. Endocrine reviews, 34(3), 309–338. https://doi.org/10.1210/er.2012-1055
- Caufriez, A., Leproult, R., L'Hermite-Balériaux, M., Kerkhofs, M., & Copinschi, G. (2011). Progesterone prevents sleep disturbances and modulates GH, TSH, and melatonin secretion in postmenopausal women. The Journal of clinical endocrinology and metabolism, 96(4), E614–E623. https://doi.org/10.1210/jc.2010-2558
- Hodis, H. N., Mack, W. J., Henderson, V. W., Shoupe, D., Budoff, M. J., Hwang-Levine, J., Li, Y., Feng, M., Dustin, L., Kono, N., Stanczyk, F. Z., Selzer, R. H., Azen, S. P., & ELITE Research Group (2016). Vascular Effects of Early versus Late Postmenopausal Treatment with Estradiol. The New England journal of medicine, 374(13), 1221–1231. https://doi.org/10.1056/NEJMoa1505241
- Heymsfield, S. B., Gonzalez, M. C., Shen, W., Redman, L., & Thomas, D. (2014). Weight loss composition is one-fourth fat-free mass: a critical review and critique of this widely cited rule. Obesity reviews : an official journal of the International Association for the Study of Obesity, 15(4), 310–321. https://doi.org/10.1111/obr.12143
- Davis, S. R., Baber, R., Panay, N., Bitzer, J., Perez, S. C., Islam, R. M., Kaunitz, A. M., Kingsberg, S. A., Lambrinoudaki, I., Liu, J., Parish, S. J., Pinkerton, J., Rymer, J., Simon, J. A., Vignozzi, L., & Wierman, M. E. (2019). Global Consensus Position Statement on the Use of Testosterone Therapy for Women. The Journal of clinical endocrinology and metabolism, 104(10), 4660–4666. https://doi.org/10.1210/jc.2019-01603
- Sánchez-Garrido, M.A., Serrano-López, V., Ruiz-Pino, F. et al. Superior metabolic improvement of polycystic ovary syndrome traits after GLP1-based multi-agonist therapy. Nat Commun 15, 8498 (2024). https://doi.org/10.1038/s41467-024-52898-y
- Khalifey, H. T., Mahereen, R., Adwan, R., Chahine, R., Kaidali, M., Mirza, S. F., Tullah, S. N., Shaikh, S., Hammad, S., & Sukkarieh, H. H. (2026). The impact of hormone replacement therapy on cardiovascular health in postmenopausal women: a narrative review. Frontiers in reproductive health, 8, 1745210. https://doi.org/10.3389/frph.2026.1745210
- Speroff L. (2010). Transdermal hormone therapy and the risk of stroke and venous thrombosis. Climacteric : the journal of the International Menopause Society, 13(5), 429–432. https://doi.org/10.3109/13697137.2010.507111
- Kim, J. J., Kurita, T., & Bulun, S. E. (2013). Progesterone action in endometrial cancer, endometriosis, uterine fibroids, and breast cancer. Endocrine reviews, 34(1), 130–162. https://doi.org/10.1210/er.2012-1043
- Goldney, J., Hamza, M., Surti, F., Davies, M. J., & Papamargaritis, D. (2025). Triple Agonism Based Therapies for Obesity. Current cardiovascular risk reports, 19(1), 18. https://doi.org/10.1007/s12170-025-00770-z
- Filippatos, T. D., Panagiotopoulou, T. V., & Elisaf, M. S. (2014). Adverse Effects of GLP-1 Receptor Agonists. The review of diabetic studies : RDS, 11(3-4), 202–230. https://doi.org/10.1900/RDS.2014.11.202
- Deutz, N. E., Bauer, J. M., Barazzoni, R., Biolo, G., Boirie, Y., Bosy-Westphal, A., Cederholm, T., Cruz-Jentoft, A., Krznariç, Z., Nair, K. S., Singer, P., Teta, D., Tipton, K., & Calder, P. C. (2014). Protein intake and exercise for optimal muscle function with aging: recommendations from the ESPEN Expert Group. Clinical nutrition (Edinburgh, Scotland), 33(6), 929–936. https://doi.org/10.1016/j.clnu.2014.04.007
- Fezza, R., Rains, B., Fezza, T., & Fezza, J. P. (2023). Emerging Anesthesia Risks with Semaglutide. Plastic and reconstructive surgery. Global open, 11(11), e5427. https://doi.org/10.1097/GOX.0000000000005427
- Giblin, K., Kaplan, L. M., Somers, V. K., Le Roux, C. W., Hunter, D. J., Wu, Q., Lalonde, A., Ahmad, N., & Bethel, M. A. (2026). Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. Diabetes, obesity & metabolism, 28(1), 83–93. https://doi.org/10.1111/dom.70209
Related studies