GLP-1 Receptor Agonists and the Hallmarks of Aging: A Systems-Level Analysis
Prefer to listen? Hit play for a conversational, audio‑style summary of this article’s key points.
Aging is driven by interacting biological processes. Cellular damage and declining repair mechanisms contribute to systemic decline, some of which may be modifiable through targeted interventions [1].
The Hallmarks of Aging framework guides intervention strategies. Primary, antagonistic, and integrative hallmarks link molecular damage to systemic aging, helping clarify where therapies like GLP-1s may have a positive influence and which physiological pathways they are most likely to influence [2,3].
GLP-1s influence metabolism, mitochondrial function, and inflammation. Studies show GLP-1 therapy can reduce reactive oxygen species (ROS), improve mitochondrial function, lower pro-inflammatory cytokines, and enhance vascular signaling, particularly in metabolic disease [5].
Effective aging strategies are combinatorial. No single therapy addresses all hallmarks. Combining pharmacologic, metabolic, and lifestyle interventions may help stabilize systems and preserve resilience.
GLP-1s support healthspan rather than reverse aging. Benefits are most evident in metabolic dysfunction, where they reduce physiological stress and complement lifestyle measures without directly modifying primary aging mechanisms.
Aging Is No Longer Just a Matter of Time
Every cell in the body constantly manages damage, repairs molecular structures, and maintains the systems required for life. Over time, these processes gradually become less efficient, leading to the accumulation of biological changes that affect metabolism, tissue function, and resilience across organ systems.
For much of modern medical history, aging was viewed primarily as an unavoidable consequence of time. Advances in molecular biology have reshaped that view. Scientists now recognize that aging reflects a set of measurable biological processes that influence how cells respond to stress, repair damage, and maintain normal function. Emerging research further suggests that these processes are not only observable but, in some cases, modifiable through targeted interventions that influence cellular signaling and systemic physiology [1].
This shift has opened a new frontier in medicine. Rather than focusing only on treating individual age-related diseases, researchers are increasingly investigating interventions that target the biological mechanisms contributing to aging itself. If these processes can be influenced, it may be possible to improve the years of life spent in good health.
Among the emerging areas of interest are therapies originally developed for metabolic disease that appear to influence multiple aging-related pathways. One notable example is the class of medications known as glucagon-like peptide-1 (GLP1) receptor agonists, which affect metabolism, inflammation, and cardiovascular health. A deeper discussion of this connection is explored in The GLP-1 Longevity Connection.
To understand why these therapies have attracted attention in longevity research, it is useful to first examine the scientific framework that describes the biological processes underlying aging.
The Hallmarks of Aging:
A Framework for Understanding Biological Aging
In 2013, López-Otín and colleagues introduced the Hallmarks of Aging framework, which was a conceptual model that organized decades of aging research into nine core biological processes that contribute to age-related decline [2]. These categories included genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication [2]. Importantly, each hallmark was defined not only by its association with aging, but also by experimental evidence suggesting that modifying these mechanisms could influence lifespan or healthspan.
Over the following decade, rapid advances in molecular biology expanded this framework. In 2023, the original authors updated the model to include twelve interconnected hallmarks of aging. This model incorporated additional processes such as chronic inflammation, dysbiosis, and impaired macroautophagy [3]. The updated model emphasizes that aging is not driven by a single mechanism, but emerges from a network of interacting biological systems that link molecular damage to systemic physiological decline.
To better understand how these processes interact, the hallmarks are often described within a three-tier hierarchy that traces the progression from molecular damage to organismal dysfunction. They include:
- Primary hallmarks, which represent the initial sources of cellular damage that accumulate over time.
- Antagonistic hallmarks, which reflect the cellular responses to this damage. Early in life, these responses help maintain cellular stability, but persistent activation over time can contribute to pathology.
- Integrative hallmarks emerge when accumulated damage and stress responses begin to overwhelm the body's repair systems.
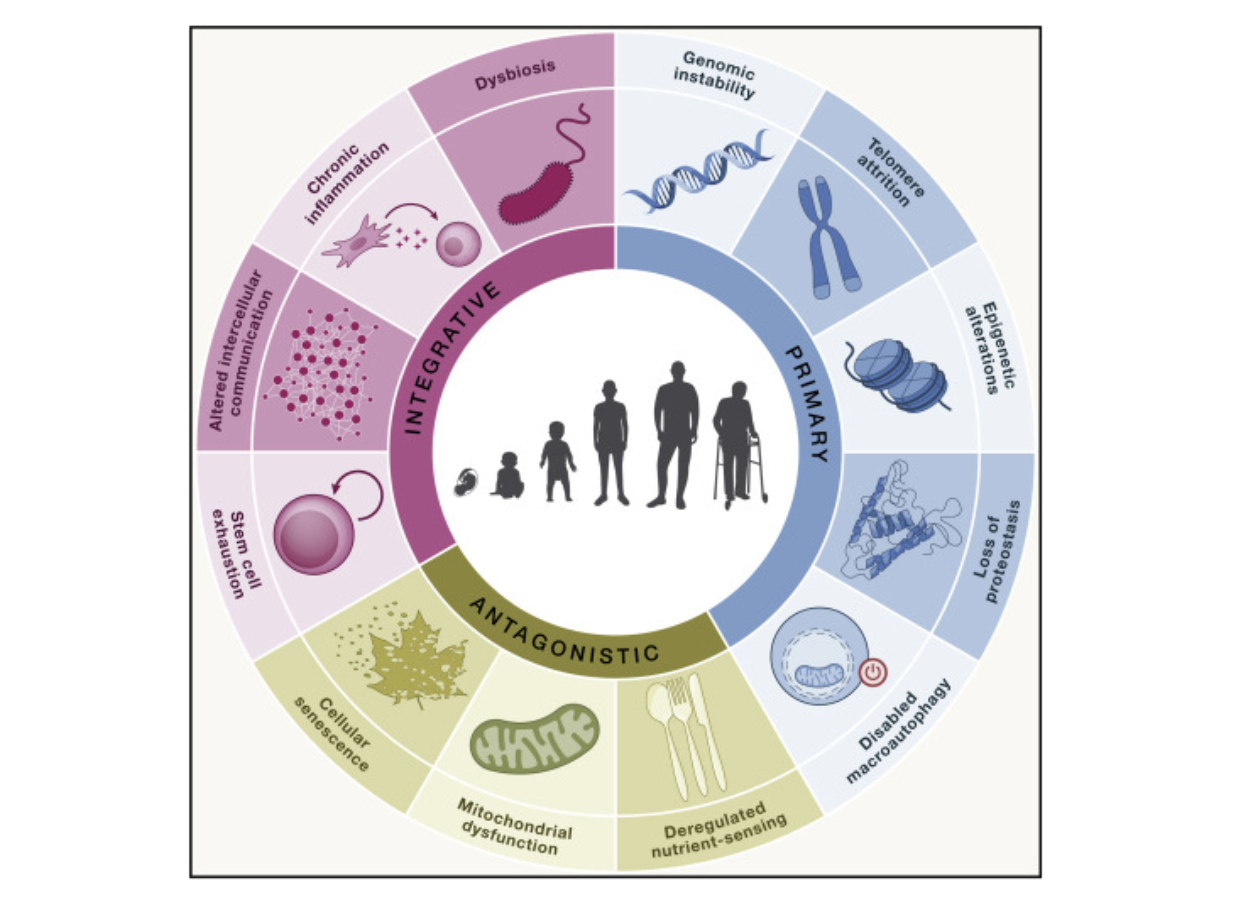
Understanding this hierarchy is important when evaluating potential geroprotective interventions. This framework offers a structured way to evaluate how emerging therapies may influence biological aging. By organizing aging into interconnected processes that span molecular damage, cellular stress responses, and systemic decline, the model helps clarify where potential interventions may exert their effects. While most current therapies do not directly address the primary drivers of molecular damage, some appear to influence downstream pathways that shape metabolic regulation, inflammatory signaling, and mitochondrial function. Among the most exciting geroprotective strategies are those that address multiple hallmarks of aging. GLP-1s, originally developed for metabolic disease, are now being investigated for their broader effects on biological systems relevant to aging.
Evidence for Anti-aging Benefits of GLP-1s
A growing body of research suggests GLP-1s have effects beyond weight loss and glycemic control. A 2023 review in Cells described these agents as promising therapies across cardiovascular, renal, neurodegenerative, and metabolic conditions, while noting that the relative contribution of direct cellular effects versus improved cardiometabolic regulation remains under investigation [4].
At the cellular level, studies suggest GLP-1 therapy may influence biological processes relevant to aging. Individuals with type 2 diabetes treated with GLP-1s have demonstrated improvements in inflammatory signaling and mitochondrial redox balance, including reduced reactive oxygen species production, improved mitochondrial membrane potential, and increased oxygen consumption. These changes were accompanied by reductions in pro-inflammatory cytokines such as IL-6 and TNF-α and increases in the anti-inflammatory cytokine IL-10 [5].
The same research also reported reduced adhesion molecules and lower carotid intima–media thickness, suggesting improved vascular regulation. These findings are encouraging, and must be interpreted cautiously, as many studies cannot separate direct pharmacologic effects from improvements in metabolic health that accompany treatment.
Additional lines of research remain more exploratory. A 2025 perspective proposed that GLP-1s could influence epigenetic pathways involved in fibrosis and hepatocellular carcinoma risk in the aging liver [6]. Experimental studies in mice have also shown that exenatide can partially reverse age-associated transcriptomic patterns in glial and neurovascular cells [7].
While these findings suggest that GLP-1s influence biological processes associated with aging, their effects are not uniformly distributed across the hallmarks framework. Most evidence to date reflects changes in downstream or adaptive pathways, such as inflammation, mitochondrial function, and metabolic regulation, rather than direct modification of the primary sources of molecular damage. This distinction is important, as it helps clarify where GLP-1 signaling may intersect with aging biology.
GLP-1 and the Primary Hallmarks of Aging
In looking first at the primary hallmarks, evidence remains limited on how GLP-1s influence these specific hallmarks. Current research primarily examines epigenetic regulation, transcriptomic aging signatures, and cellular quality-control systems. Let’s take a look at some of the early findings.
Epigenetic and Transcriptomic Regulation
A 2021 study in Communications Biology reported that treatment with the GLP-1s exenatide reversed transcriptomic aging signatures across multiple mouse brain cell populations, including glial and neurovascular cells [7]. The most pronounced effects were observed in glial cells, the metabolic and inflammatory support system of the brain, suggesting that improving the brain’s environment may be as important as targeting neurons directly.
Although biologically notable, transcriptomic shifts do not necessarily indicate tissue rejuvenation or restored function in humans. Rather, they reflect changes in gene expression patterns, indicating which genes are activated or suppressed, and to what extent. Without confirming functional outcomes at the tissue or organism level.
A related hypothesis has been proposed in hepatic aging. A 2025 perspective suggested GLP-1s may influence epigenetic processes involved in age-related liver fibrosis and cancer risk [6].
Cellular Quality Control and Glycation Stress
Macroautophagy, a system that removes damaged proteins and organelles, also plays a role in cellular aging. Metabolic stress promotes the formation of advanced glycation end products (AGEs), which activate inflammatory signaling through the receptor for advanced glycation end products (RAGE) [8].
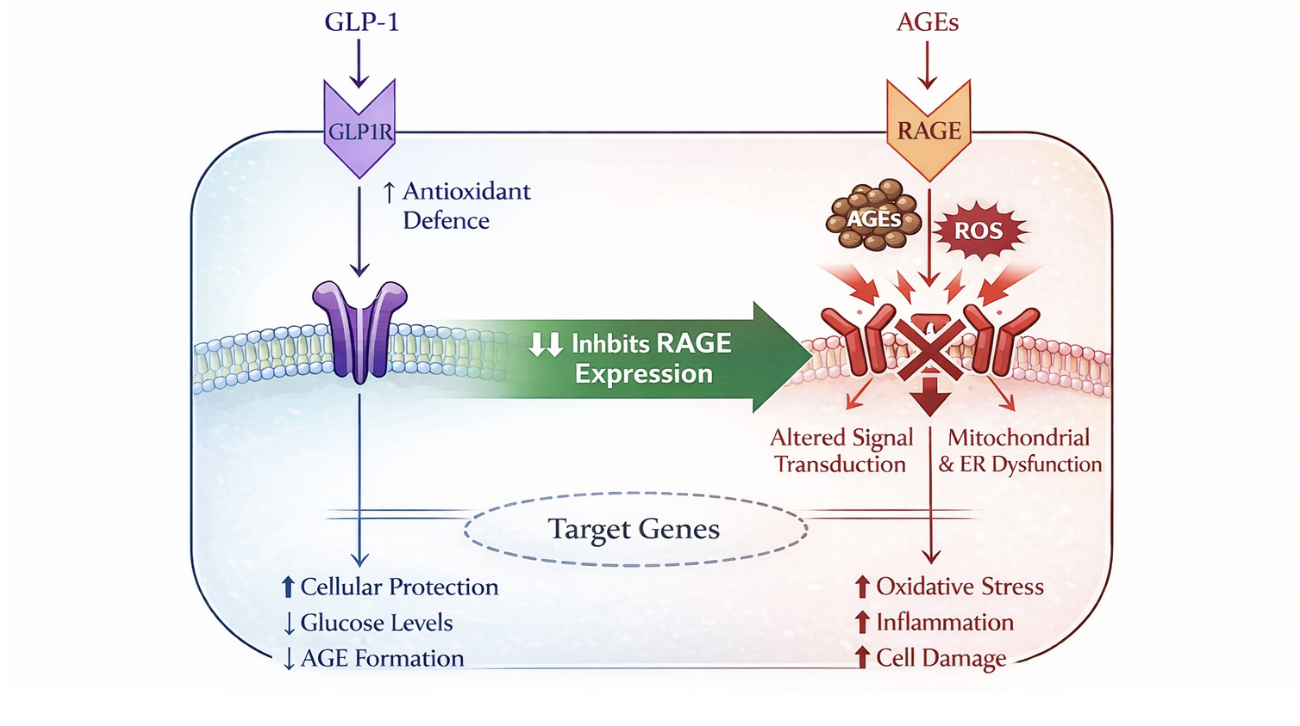
Figure 2. GLP-1 Modulation of AGEs via RAGE Signaling
In a 2024 cell biology study, the GLP-1 receptor agonist liraglutide reduced inflammation and apoptosis in AGE-exposed chondrocytes by inhibiting RAGE signaling [9]. This included a reduction in RAGE protein expression, suppression of key inflammatory cytokines (IL-6, IL-12, TNF-α), and attenuation of matrix-degrading enzymes [9]. These changes were accompanied by preservation of structural proteins including aggrecan and collagen II, along with reduced caspase-3–mediated apoptosis, suggesting a coordinated reduction in glycation-driven cellular stress and tissue degradation. These findings suggest a potential mechanism through which GLP-1 signaling may reduce glycation-related cellular stress.
Interpreting the Evidence
Across studies, these findings suggest that GLP-1s may influence biological processes associated with primary hallmarks, particularly epigenetic regulation, inflammatory signaling, and cellular stress responses. However, the strongest evidence currently comes from models of metabolic dysfunction.
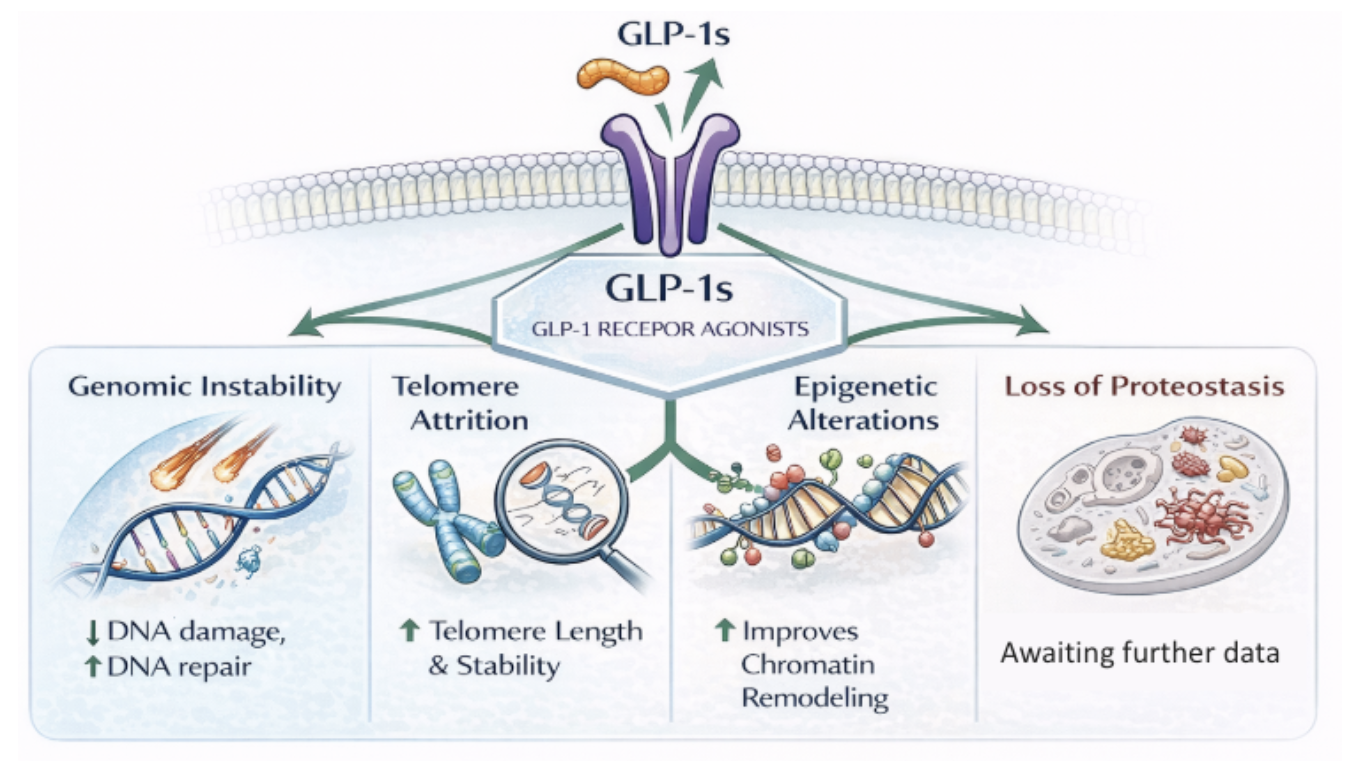
Figure 3. GLP1 Impacts on Primary Hallmarks of Aging
Importantly, this does not imply direct modification of primary aging processes such as genomic instability or telomere attrition. Instead, by improving metabolic regulation and reducing chronic inflammatory stress, GLP-1 signaling may alter the cellular environment in ways that support more effective management of accumulated molecular damage.
For this reason, the most consistent connections between GLP-1 biology and aging emerge in the next tier of the hallmarks of aging framework, Antagonistic. This is especially evident in mitochondrial dysfunction, which links nutrient sensing, energy production, and inflammatory stress within a single, highly integrated system.
GLP-1s and Antagonistic Hallmarks: Mitochondrial Dysfunction and Redox Regulation
Mitochondrial dysfunction often emerges downstream of disrupted nutrient sensing and chronic metabolic stress, linking cellular energetics to broader aging biology. Beyond ATP production, mitochondria regulate redox balance and cellular stress responses that are essential for maintaining tissue function over time. With aging, impairments in mitochondrial efficiency are associated with increased reactive oxygen species (ROS) production and declining bioenergetic capacity [10].
In the Redox Biology study described earlier [5], individuals with type 2 diabetes treated with GLP-1 receptor agonists demonstrated reduced ROS production, restoration of mitochondrial membrane potential, improved oxygen consumption, and lower levels of myeloperoxidase (MPO). These findings suggest improved mitochondrial energetics and redox regulation within immune and vascular compartments.
Mechanistically, these effects are coherent. Enhanced mitochondrial efficiency can reduce oxidative stress and improve cellular energy availability. However, it remains uncertain whether these observations reflect direct modulation of aging-related mitochondrial decline or secondary improvements resulting from restored metabolic homeostasis.
Dysregulated Nutrient Sensing
Mitochondrial function is tightly integrated with nutrient-sensing pathways, another antagonistic hallmark governing how cells respond to energy availability. Signaling networks involving insulin, AMP-activated protein kinase (AMPK), and mTOR coordinate cellular growth, repair, and energy utilization, and become progressively dysregulated with age.
GLP-1 receptor agonists appear to influence several components of this system.
Experimental and clinical data suggest increased AMPK activity, improved insulin sensitivity, and modulation of mTOR-related signaling, patterns that overlap with metabolic adaptations observed during caloric restriction [11,12]. These pathways play a central role in mitochondrial biogenesis, energy efficiency, and cellular stress resistance.
This overlap provides a biologically plausible framework through which GLP-1 signaling may help stabilize cellular energy regulation, particularly in the context of metabolic stress, even if direct effects on intrinsic aging processes remain to be fully established.
Cellular Senescence
Mitochondrial dysfunction and persistent metabolic stress are key contributors to cellular senescence, a state characterized by loss of replicative capacity and altered cellular signaling. While senescent cells are often discussed in the context of inflammatory signaling, their accumulation is fundamentally linked to impaired energy metabolism and oxidative stress.
Preclinical studies suggest that GLP-1 signaling may reduce oxidative stress markers and modulate pathways associated with cellular stress responses in vascular and metabolic tissues [13,14]. Although this does not indicate direct senolytic activity, it is consistent with a model in which improved mitochondrial function and nutrient signaling reduce the upstream conditions that promote senescence.
In this context, GLP-1 therapies may act less by eliminating senescent cells and more by slowing the processes that drive their accumulation over time.
Interpreting the Evidence
GLP-1 receptor agonists intersect with several antagonistic hallmarks of aging, particularly those linking mitochondrial function, nutrient sensing, and cellular senescence.
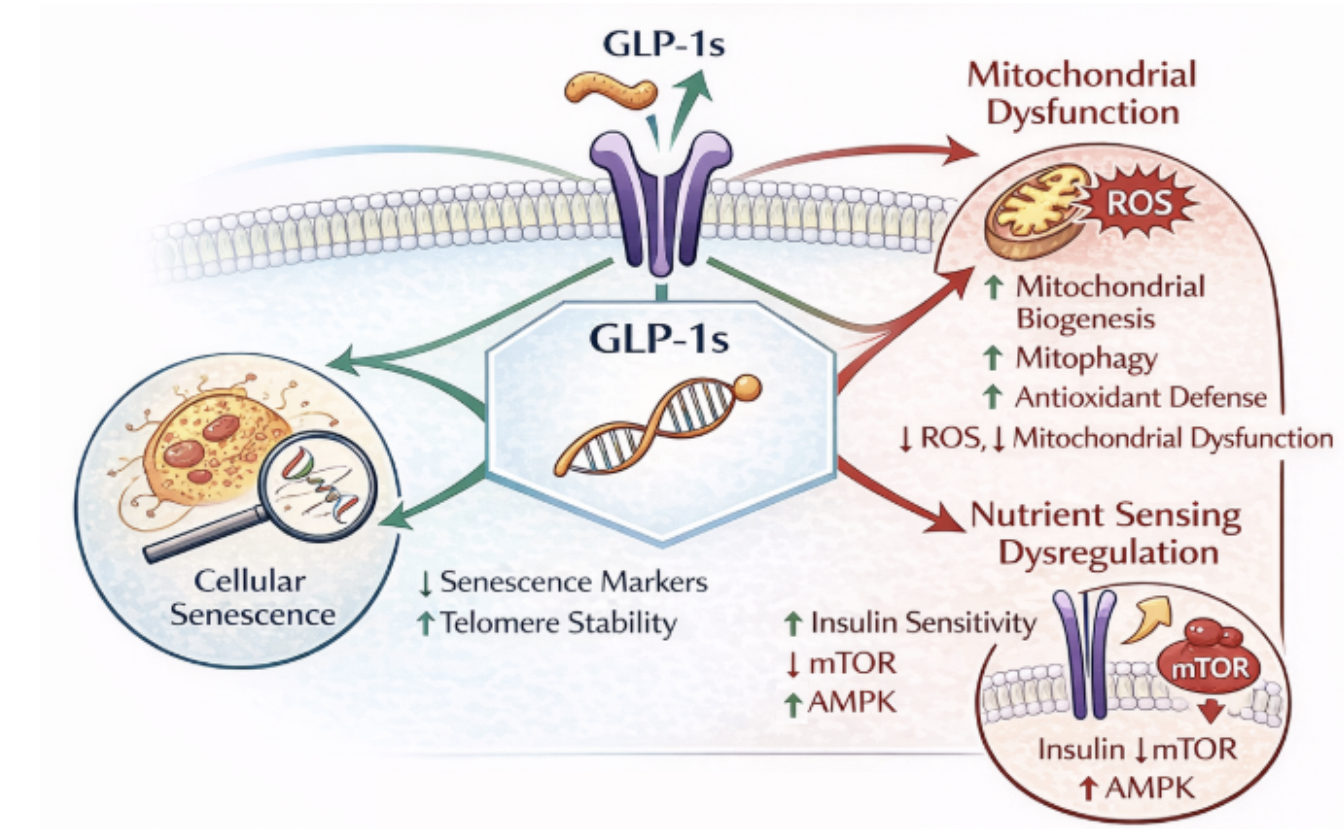
Figure 4. GLP1 Impacts on Antagonistic Hallmarks of Aging
However, much of the current evidence derives from populations with metabolic disease, animal models, or cellular systems. As a result, it remains difficult to distinguish whether these effects represent direct modification of aging biology or improvements in underlying metabolic dysfunction.
Even so, these pathways are central to maintaining cellular and tissue function over time. Interventions that support mitochondrial efficiency, stabilize nutrient sensing, and limit the accumulation of senescent cells may contribute to preserving physiological resilience, even if they do not directly alter the primary drivers of aging.
GLP-1s and the Integrative Hallmarks of Aging: Inflammaging and Systemic Signaling
Chronic inflammation, or inflammaging, is a central integrative hallmark of aging. Unlike acute inflammation, it reflects a persistent, low-grade, non-resolving state that increases over time and is associated with age-related disease and mortality [10].
This inflammatory state extends beyond the immune system. Inflammatory signaling influences metabolic regulation, endothelial function, mitochondrial activity, and tissue repair. Over time, these interactions form reinforcing feedback loops that amplify physiologic stress across organ systems. Because GLP-1s affect metabolic signaling, insulin sensitivity, and inflammatory pathways, their potential influence on this broader inflammatory environment has been examined.
A 2023 study in Redox Biology evaluated inflammatory and vascular signaling in individuals with type 2 diabetes treated with GLP-1s for at least one year (n=59), compared with untreated patients (n=196) and healthy controls (n=175) [15].
Compared with untreated individuals, those receiving GLP-1 therapy showed lower levels of pro-inflammatory cytokines (IL-6, TNF-α, IL-12), higher levels of IL-10, reduced expression of endothelial adhesion molecules (ICAM-1, VCAM-1), and decreased leukocyte rolling and adhesion along the vascular endothelium [15].
These findings indicate a shift toward lower inflammatory signaling and improved vascular–immune regulation. Reduced endothelial activation and leukocyte adhesion are particularly relevant to early vascular dysfunction. However, the study was cross-sectional, demonstrating association rather than causation.
Altered Intercellular Communication and Immune–Endothelial Cross-Talk
Aging is also characterized by altered intercellular communication, particularly between immune cells, vascular tissue, and metabolic organs. With metabolic stress, leukocyte–endothelial interactions become more adhesive and pro-inflammatory, contributing to endothelial dysfunction, vascular stiffness, and atherosclerotic progression.
In the same Redox Biology study, GLP-1–treated patients exhibited increased leukocyte rolling velocity and reduced adhesion compared with untreated individuals [15]. These changes occurred alongside reductions in inflammatory cytokines and adhesion molecules.
This pattern is consistent with reduced inflammatory signaling at the vascular interface and fewer cues for sustained endothelial activation, suggesting improved vascular stability.
Interpreting the Evidence
Much like with the Antagonistic hallmarks, current evidence in improvements of Integrative Hallmarks are strongest in populations with metabolic dysfunction, particularly type 2 diabetes. While these findings align with mechanisms relevant to aging biology, they do not establish that GLP-1 therapies directly modify intrinsic aging processes.
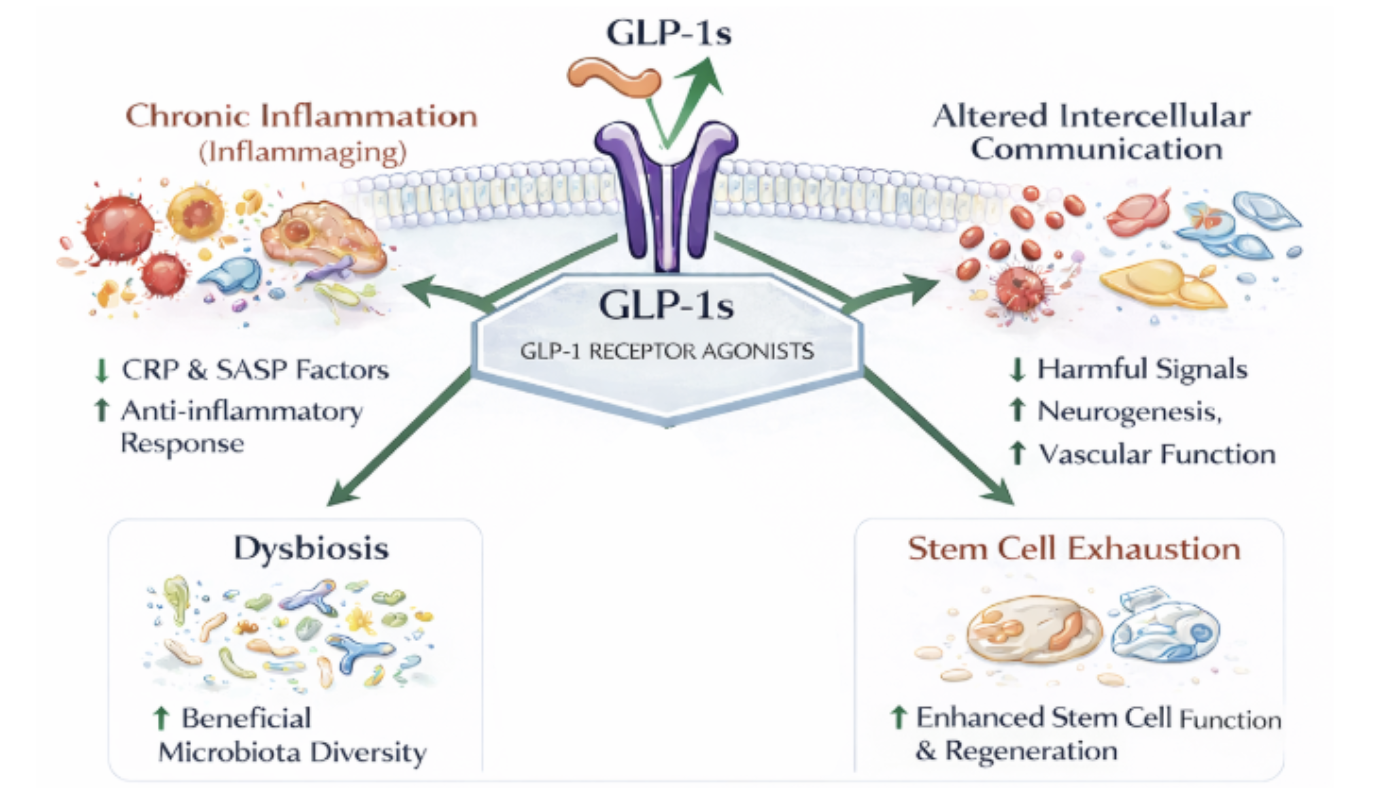
Figure 5. GLP1 Impacts on Integrative Hallmarks of Aging
Instead, the data suggest that GLP-1s may reduce systemic physiological stress, particularly where metabolic dysfunction, vascular inflammation, and immune signaling intersect. By stabilizing these interconnected systems, such interventions may support metabolic resilience, vascular function, and overall physiological integrity over time.
Emerging Research: Combination Strategies for the Hallmarks of Aging
While GLP-1s have attracted attention for their metabolic and cardiometabolic effects, aging biology suggests that no single intervention is likely to address all hallmarks simultaneously. Aging reflects a network of interacting processes, and current research increasingly emphasizes combinatorial strategies that integrate pharmacologic, metabolic, and lifestyle-based interventions. Several emerging lines of evidence illustrate how different approaches may influence distinct hallmarks.
For example, recent work on SGLT2 inhibitors suggests potential effects on telomere biology. A 2025 study reported evidence of telomere length stabilization or modest lengthening, raising the possibility that metabolic therapies may influence pathways related to genomic stability and replicative lifespan [16]. Although early, these findings highlight how therapies developed for cardiometabolic disease may intersect with hallmarks such as telomere attrition.
Other investigations focus on cellular senescence. Rapamycin and related mTOR pathway modulators are being studied for their effects on senescence-associated signaling. A 2025 preclinical study reported modulation of senescence-related pathways consistent with improved cellular function, though translation to human aging remains uncertain [17].
Not all interventions are pharmacologic. Lifestyle-based strategies also influence hallmark pathways, too. A 2021 clinical study examining a combined diet and lifestyle program reported an average 3.23-year reduction in DNA methylation age (DNAmAge) [18]. These findings suggest that coordinated changes in nutrition, sleep, and stress regulation may affect epigenetic aging programs.
These examples point toward a consistent theme. Different interventions appear to act on distinct, but overlapping biological pathways. Metabolic therapies such as GLP-1s primarily influence inflammation, mitochondrial function, and nutrient signaling, while other pharmacological or lifestyle strategies may act on telomere maintenance [16], senescence biology [17], or epigenetic regulation [18].
In this context, GLP-1 therapies are best understood as one component within a broader strategy. When combined with interventions that support metabolic resilience, cellular maintenance, and behavioral stability, they may contribute to maintaining physiological function over time. As the field evolves, understanding how these approaches interact will be central to developing durable strategies for healthy aging.
Where GLP-1s Fit in a Healthspan Strategy
GLP-1s should not be framed as “anti-aging” drugs. Rather, they are tools to reduce key drivers of physiological decline while supporting foundational systems. When clinically appropriate, GLP-1 therapy can help lower cardiometabolic strain and inflammatory burden while complementing, not replacing, lifestyle fundamentals such as nutrient-dense eating, regular movement, resistance training, adequate protein intake, and restorative sleep.
The benefits of GLP-1 therapy are context-dependent, with the clearest signals observed in individuals experiencing metabolic dysfunction. In these populations, therapy can help stabilize appetite regulation, glycemic control, and inflammatory signaling. Use should always occur under clinical supervision with individualized evaluation, monitoring, and follow-up.
Who Might Benefit Most?
Evidence suggests the strongest effects appear in individuals with elevated metabolic burden. In these settings, glucose dysregulation, visceral adiposity, fatty liver, vascular inflammation, and chronic low-grade inflammation often reinforce one another, creating a cascade of metabolic stress that accelerates functional decline.
By improving nutrient signaling and reducing appetite dysregulation, GLP-1 therapies may lower stress across multiple organ systems. Populations most likely to benefit include individuals with obesity or central adiposity, insulin resistance or type 2 diabetes, metabolic dysfunction–associated fatty liver disease, or elevated cardiovascular risk. In such cases, lowering metabolic and inflammatory stress pathways can help preserve long-term physiological resilience.
It is important to emphasize that GLP-1 therapies do not directly modify fundamental aging mechanisms. Their value lies in situations where metabolic dysfunction actively contributes to age-related decline.
Conclusion
GLP-1 receptor agonists function as multi-system metabolic modulators that influence pathways intersecting with aging biology. By improving metabolic regulation, moderating nutrient signaling, and reducing chronic inflammatory burden, these therapies may help alleviate physiological stress that contributes to age-related disease and functional decline.
Within the Hallmarks of Aging framework, their effects are most evident across metabolic, inflammatory, and mitochondrial pathways that connect cellular stress to systemic dysfunction. This does not indicate direct modification of primary aging mechanisms such as genomic instability or telomere attrition. Rather, GLP-1 therapies appear to support the biological environment in which these processes unfold.
Future research incorporating validated biomarkers of biological aging, functional outcomes, and long-term disease risk will clarify their role more precisely. In the meantime, GLP-1s represent a clinically relevant tool for improving metabolic resilience, reducing systemic inflammatory stress, and supporting long-term physiological function when applied appropriately.
- Kroemer, G., Maier, A. B., Cuervo, A. M., Gladyshev, V. N., Ferrucci, L., Gorbunova, V., Kennedy, B. K., Rando, T. A., Seluanov, A., Sierra, F., Verdin, E., & López-Otín, C. (2025). From geroscience to precision geromedicine: Understanding and managing aging. Cell, 188(8), 2043–2062. https://doi.org/10.1016/j.cell.2025.03.011
- López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2013). The hallmarks of aging. Cell, 153(6), 1194–1217. https://doi.org/10.1016/j.cell.2013.05.039
- López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2023). Hallmarks of aging: An expanding universe. Cell, 186(2), 243–278. https://doi.org/10.1016/j.cell.2022.11.001
- Wilbon, S. S., & Kolonin, M. G. (2023). GLP1 Receptor Agonists-Effects beyond Obesity and Diabetes. Cells, 13(1), 65. https://doi.org/10.3390/cells13010065
- Luna-Marco, C., de Marañon, A. M., Hermo-Argibay, A., Rodriguez-Hernandez, Y., Hermenejildo, J., Fernandez-Reyes, M., Apostolova, N., Vila, J., Sola, E., Morillas, C., Rovira-Llopis, S., Rocha, M., & Victor, V. M. (2023). Effects of GLP-1 receptor agonists on mitochondrial function, inflammatory markers and leukocyte-endothelium interactions in type 2 diabetes. Redox biology, 66, 102849. https://doi.org/10.1016/j.redox.2023.102849
- Hanna, S., Sethiadi, J., Ali, Q., & Sinha, S. (2025). Repurposing GLP-1 Receptor Agonists: A Perspective on Epigenetic Strategies to Combat Fibrosis and Hepatocellular Carcinoma in the Aged Liver. Cancers, 17(16), 2600. https://doi.org/10.3390/cancers17162600
- Li, Z., Chen, X., Vong, J. S. L., Zhao, L., Huang, J., Yan, L. Y. C., Ip, B., Wing, Y. K., Lai, H. M., Mok, V. C. T., & Ko, H. (2021). Systemic GLP-1R agonist treatment reverses mouse glial and neurovascular cell transcriptomic aging signatures in a genome-wide manner. Communications biology, 4(1), 656. https://doi.org/10.1038/s42003-021-02208-9
- Vianello, E., Beltrami, A. P., Aleksova, A., Janjusevic, M., Fluca, A. L., Corsi Romanelli, M. M., La Sala, L., & Dozio, E. (2025). The Advanced Glycation End-Products (AGE)-Receptor for AGE System (RAGE): An Inflammatory Pathway Linking Obesity and Cardiovascular Diseases. International journal of molecular sciences, 26(8), 3707. https://doi.org/10.3390/ijms26083707
- Zhang, X., Jiang, J., Xu, J., Chen, J., Gu, Y., & Wu, G. (2024). Liraglutide, a glucagon-like peptide-1 receptor agonist, ameliorates inflammation and apoptosis via inhibition of receptor for advanced glycation end products signaling in AGEs induced chondrocytes. BMC musculoskeletal disorders, 25(1), 601. https://doi.org/10.1186/s12891-024-07640-6
- Baechle, J. J., Chen, N., Makhijani, P., Winer, S., Furman, D., & Winer, D. A. (2023). Chronic inflammation and the hallmarks of aging. Molecular metabolism, 74, 101755. https://doi.org/10.1016/j.molmet.2023.101755
- Müller, T. D., Finan, B., Bloom, S. R., D'Alessio, D., Drucker, D. J., Flatt, P. R., Fritsche, A., Gribble, F., Grill, H. J., Habener, J. F., Holst, J. J., Langhans, W., Meier, J. J., Nauck, M. A., Perez-Tilve, D., Pocai, A., Reimann, F., Sandoval, D. A., Schwartz, T. W., Seeley, R. J., … Tschöp, M. H. (2019). Glucagon-like peptide 1 (GLP-1). Molecular metabolism, 30, 72–130. https://doi.org/10.1016/j.molmet.2019.09.010
- Drucker D. J. (2018). Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell metabolism, 27(4), 740–756. https://doi.org/10.1016/j.cmet.2018.03.001
- Bendotti, G., Montefusco, L., Lunati, M. E., Usuelli, V., Pastore, I., Lazzaroni, E., Assi, E., Seelam, A. J., El Essawy, B., Jang, J., Loretelli, C., D'Addio, F., Berra, C., Ben Nasr, M., Zuccotti, G., & Fiorina, P. (2022). The anti-inflammatory and immunological properties of GLP-1 Receptor Agonists. Pharmacological research, 182, 106320. https://doi.org/10.1016/j.phrs.2022.106320
- Hogan, A. E., Davis, C., Jenkins, B. J., Jones, N., & O'Shea, D. (2025). Repurposing metabolic drugs as anti-inflammatory agents. Trends in endocrinology and metabolism: TEM, S1043-2760(25)00149-3. Advance online publication. https://doi.org/10.1016/j.tem.2025.07.003
- Luna-Marco, C., de Marañon, A. M., Hermo-Argibay, A., Rodriguez-Hernandez, Y., Hermenejildo, J., Fernandez-Reyes, M., Apostolova, N., Vila, J., Sola, E., Morillas, C., Rovira-Llopis, S., Rocha, M., & Victor, V. M. (2023). Effects of GLP-1 receptor agonists on mitochondrial function, inflammatory markers and leukocyte-endothelium interactions in type 2 diabetes. Redox biology, 66, 102849. https://doi.org/10.1016/j.redox.2023.102849
- Zhang, J., Cai, W., Liu, D., Zheng, N., Wang, Y., Qiu, F., Zheng, H., Gan, H., Huang, Y., Zhou, Y., Yu, M., Xiong, S., Luo, G., Guo, J., Zhu, L., Zhang, Y., Ke, H., Liu, Y., Huang, G., Yu, C., … Xu, J. (2025). Effect of henagliflozin on aging biomarkers in patients with type 2 diabetes: A multicenter, randomized, double-blind, placebo-controlled study. Cell reports. Medicine, 6(9), 102331. https://doi.org/10.1016/j.xcrm.2025.102331
- Kell, L., Jones, E. J., Gharahdaghi, N., Wilkinson, D. J., Smith, K., Atherton, P. J., Simon, A. K., Cox, L. S., & Alsaleh, G. (2026). Rapamycin Exerts Its Geroprotective Effects in the Ageing Human Immune System by Enhancing Resilience Against DNA Damage. Aging cell, 25(2), e70364. https://doi.org/10.1111/acel.70364
- Fitzgerald, K. N., Hodges, R., Hanes, D., Stack, E., Cheishvili, D., Szyf, M., Henkel, J., Twedt, M. W., Giannopoulou, D., Herdell, J., Logan, S., & Bradley, R. (2021). Potential reversal of epigenetic age using a diet and lifestyle intervention: a pilot randomized clinical trial. Aging, 13(7), 9419–9432. https://doi.org/10.18632/aging.202913
Related studies