Widespread Oxytocin Receptor Expression Suggests a Broader Role in Longevity Biology
Oxytocin functions as a multisystem regulatory hormone. Oxytocin receptors are widely distributed across the brain, cardiovascular system, bone, adipose tissue, and gastrointestinal tract, allowing it to influence multiple systems involved in healthspan.
Oxytocin helps buffer the stress response. Human studies show oxytocin can reduce peak cortisol responses to acute stress by roughly 10–30%, supporting faster recovery from sympathetic “fight-or-flight” activation.
Oxytocin influences mood, anxiety, and social cognition. Neuroimaging research demonstrates 20–40% reductions in amygdala threat responses and 10–25% improvements in anxiety scores, alongside increased trust and social engagement.
Sleep architecture may improve with oxytocin signaling. Studies report improvements across multiple sleep parameters including 10–20 minute reductions in sleep latency, 5–10% improvements in sleep efficiency, and 10–15% increases in REM sleep episodes.
Oxytocin may influence appetite and boost bone health. Experimental feeding studies show 10–15% reductions in caloric intake and have been shown to boost bone formation activity by roughly 30–40% in experimental models.
Mode of delivery shapes oxytocin’s effects. Because oxytocin has a relatively short half-life, pharmacokinetics and route of administration meaningfully influence outcomes.
Responses are individualized and work best within a broader health framework. Optimal outcomes typically involve personalized dosing, intermittent use, and supportive lifestyle practices such as sleep optimization, exercise, and social connection.
Oxytocin as a Multisystem Regulator of Healthspan
Oxytocin is often described as the body’s “bonding hormone,” historically recognized for its roles in childbirth, lactation, and maternal attachment. Over the past two decades, however, research has expanded this view dramatically. Oxytocin is now understood to function as a widely distributed neuromodulator that influences stress regulation, emotional processing, metabolic signaling, and autonomic nervous system balance [1,2].
Oxytocin-producing neurons originate in the hypothalamic paraventricular and supraoptic nuclei and project throughout the brain and body, allowing this peptide hormone to influence multiple physiological systems simultaneously [1]. Oxytocin receptors are expressed not only in regions involved in emotional regulation, but also in peripheral tissues, including the cardiovascular system, bone, adipose tissue, and gastrointestinal tract [3-5]. This broad receptor distribution suggests oxytocin may function less like a single-purpose hormone and more like a regulatory signal coordinating diverse physiological processes throughout the body.
Early studies suggest potential effects on stress resilience, mood regulation, sleep architecture, appetite control, bone remodeling, and pain perception [1,2]. While the clinical literature is still developing, the breadth of physiologic systems influenced by oxytocin has generated growing interest in its potential role in supporting quality of life and long-term healthspan.
In this review, we examine the emerging science surrounding oxytocin as a regulatory hormone influencing stress physiology, sleep, mood, metabolic signaling, skeletal health, and pain perception. We also explore delivery strategies, individual variability in response, and the lifestyle factors that naturally support oxytocin signaling.
Stress Regulation and Cortisol Modulation
One of the most extensively studied roles of oxytocin involves its interaction with the body’s primary stress-response network, the hypothalamic–pituitary–adrenal (HPA) axis. Oxytocin-producing neurons in the hypothalamus project to brain regions including the amygdala and hippocampus, which evaluate threat, emotional salience, and physiological stress responses [1]. Through these pathways, oxytocin signaling can dampen excessive HPA-axis activation, moderating cortisol release and helping the body transition from a sympathetic “fight-or-flight” state toward parasympathetic recovery [6].
Mechanistically, oxytocin influences both central and peripheral stress signaling. Within the amygdala, it reduces neural reactivity to perceived threats, while interactions with the hippocampus and hypothalamus help recalibrate emotional processing and stress memory [6]. These effects may help end chronic stress cycles and prolonged cortisol elevation following acute stress, a pattern increasingly associated with metabolic dysfunction, cardiovascular disease, or mood disorders when sustained over time [7].
Controlled human studies support this regulatory role. A meta-analysis examining oxytocin’s effects on stress responses reported reductions in cortisol reactivity during experimentally induced stress tasks such as the Trier Social Stress Test [6]. Across several studies, intranasal oxytocin has been shown to reduce peak cortisol responses to acute stress by roughly 10–30% compared with placebo, particularly when administered before stress exposure [6,7].
More recent research has explored intranasal oxytocin’s ability to influence stress physiology in humans by accessing central nervous system pathways through olfactory and trigeminal routes. Emerging studies report measurable effects on stress perception, emotional reactivity, autonomic balance, and cortisol recovery, although responses vary depending on baseline stress levels, social context, and individual receptor sensitivity [1,2,6].
Oxytocin may function as a counterbalance hormone during times of stress. By moderating cortisol output and improving adaptive stress responses, oxytocin signaling may help restore regulatory balance across multiple physiological systems involved in long-term healthspan.
Across several studies, intranasal oxytocin has been shown to reduce peak cortisol responses to acute stress by roughly 10–30%
Mood Regulation, Anxiety, Social Connectivity, and Social Behavior
Beyond its role in stress physiology, oxytocin has been widely studied for its influence on social-emotional regulation. Its effects emerge not from a single brain region, but from coordinated activity across a distributed network that includes the amygdala, hippocampus, prefrontal cortex, and hypothalamus—areas that collectively shape how we interpret social signals, evaluate potential threats, and generate emotional responses.
Within this circuitry, oxytocin appears to recalibrate the brain’s sensitivity to social information. The amygdala, which plays a central role in detecting threat and assigning emotional salience, often shows reduced activation in response to negative or fear-inducing stimuli under the influence of oxytocin. At the same time, regions of the prefrontal cortex involved in top-down regulation of emotion become more engaged, suggesting a shift toward more controlled and context-appropriate responses. In practical terms, this can translate into a heightened ability to attend to positive social cues—such as facial expressions of trust or affiliation—while dampening excessive reactivity within limbic stress pathways [1,6,9].
These limbic stress pathways refer to a set of evolutionarily conserved circuits centered around the amygdala and its connections to the hypothalamus and brainstem. When activated, this network helps initiate the body’s stress response—triggering the release of cortisol, increasing sympathetic nervous system activity, and preparing the organism for “fight-or-flight.” While this response is essential for survival in acute situations, chronic overactivation can lead to heightened anxiety, exaggerated threat perception, and impaired emotional regulation. By modulating activity within these circuits, oxytocin may help shift the balance away from persistent threat vigilance and toward a more regulated, socially attuned state.
Oxytocin’s influence extends further through its interactions with key neurotransmitter systems that govern mood and motivation. It modulates serotonergic pathways, which are closely tied to emotional stability and anxiety regulation, as well as dopaminergic circuits involved in reward processing and reinforcement learning. This dual interaction may help explain why oxytocin is associated not only with reduced stress reactivity, but also with enhanced feelings of social connection and reinforcement of bonding behaviors [8,9]. In this way, oxytocin functions less as a simple “bonding hormone” and more as a neuromodulator that fine-tunes how the brain prioritizes and responds to the social world.
Experimental and clinical studies suggest oxytocin can produce measurable effects on anxiety and emotional regulation. Intranasal oxytocin has been shown to reduce amygdala activation to threatening stimuli by roughly 20–40% in functional neuroimaging studies, while simultaneously increasing activity in prefrontal regions involved in emotional control [8]. Behavioral studies also report reductions in self-reported anxiety scores of approximately 10–25% in controlled laboratory settings, particularly during social stress or threat-evaluation tasks [10]. These findings have prompted investigation of oxytocin as a potential adjunct therapy for conditions such as generalized anxiety, depression, post-traumatic stress disorder, and social anxiety, where dysregulated threat processing and impaired social signaling play central roles [9].
Oxytocin’s influence extends beyond mood into broader domains of social connectivity and affiliative behavior. Human studies demonstrate that oxytocin administration can increase measures of interpersonal trust by roughly 15–20%, improve recognition of emotional facial expressions, and strengthen social memory and empathic processing [10]. These effects appear particularly relevant in contexts involving parenting, caregiving, and romantic attachment, where oxytocin reinforces social reward circuitry and cooperative behavior. Reviews of the oxytocin literature therefore describe it as a key neuromodulator of social salience, helping individuals interpret and respond to social cues more adaptively [8,10].
These behavioral effects also intersect with broader physiological health outcomes. Chronic anxiety, social isolation, and impaired emotional regulation are associated with elevated sympathetic tone, reduced heart rate variability, disrupted sleep, and increased cardiometabolic risk [8-10]. By improving emotional regulation and strengthening social signaling pathways, oxytocin may indirectly support autonomic balance, stress recovery, and long-term resilience. Although responses vary across individuals, the breadth of evidence suggests oxytocin occupies a unique position at the intersection of emotional health, social behavior, and physiological regulation.
Sleep Regulation
Sleep is another physiological domain where oxytocin signaling may play a meaningful regulatory role. Oxytocin influences neural pathways involved in circadian rhythm stability, autonomic balance, and emotional calm factors that contribute to both sleep initiation and sleep quality [1,2]. By interacting with stress-related brain regions and promoting parasympathetic activity, oxytocin may help shift the body away from hyper-arousal states that interfere with normal sleep onset and nighttime recovery [2].
Several lines of research suggest that oxytocin’s stress-buffering effects can translate into measurable improvements in sleep physiology [7,10]. Because oxytocin dampens excessive HPA-axis activity and supports emotional regulation, individuals may experience shorter sleep onset latency, fewer nighttime awakenings, and improved sleep efficiency. In controlled human studies, intranasal oxytocin administration has been associated with reductions in sleep latency of roughly 10–20 minutes and increases in overall sleep efficiency of approximately 5–10%, suggesting improvements in the transition from wakefulness to stable sleep states [2,11].
Experimental studies provide further insight into oxytocin’s influence on sleep architecture. In one human investigation, oxytocin administration was associated with increases in REM sleep episodes of approximately 10–15%, along with improved sleep efficiency and faster sleep onset [2,11]. REM sleep plays a central role in emotional regulation and memory consolidation, and emerging evidence suggests oxytocin signaling during REM may enhance aspects of emotional memory processing and cognitive integration during sleep cycles [12].
Interest in oxytocin and sleep also extends to aging populations. Endogenous oxytocin levels decline with age, a pattern that parallels the increasing prevalence of sleep disturbances later in life [1,2]. While clinical research remains limited, investigators have proposed that oxytocin supplementation may represent a potential therapeutic strategy for improving sleep, especially in midlife women [2] as well as individuals of advancing age by supporting stress recovery, autonomic balance, and circadian stability [11].
From a healthspan perspective, oxytocin’s potential influence on sleep is particularly relevant because sleep architecture supports multiple systems and routine recovery. High-quality sleep promotes glymphatic clearance of metabolic waste from the brain, regulates metabolic and hormonal signaling, and protects against neurodegenerative processes [11,12]. By stabilizing stress physiology and autonomic tone, oxytocin signaling may help support more resilient sleep patterns and downstream physiological recovery over time.
oxytocin administration was associated with increases in REM sleep episodes of approximately 10–15%, along with improved sleep efficiency and faster sleep onset
Appetite Regulation and Metabolic Signaling
Oxytocin also appears to influence hypothalamic feeding circuits that regulate appetite, satiety, and food reward processing. Oxytocin receptors are expressed in brain regions involved in energy balance, including the hypothalamus, ventral tegmental area, and nucleus accumbens [2,13]. Through these pathways, oxytocin signaling can influence both homeostatic hunger signals and hedonic eating behaviors, helping regulate caloric intake and reward-driven food consumption [13,14].
Human feeding studies provide measurable evidence for these effects. In controlled laboratory experiments, intranasal oxytocin administration has been shown to reduce caloric intake during test meals by approximately 10–15% (roughly 70-100 kcal for an average meal around 700 total kcal), with particularly pronounced reductions in high-sugar snack consumption and reward-driven eating [13]. In one randomized crossover study involving healthy men, oxytocin reduced snack intake by roughly 20–25% during post-meal snack exposure, suggesting that oxytocin may specifically influence hedonic eating rather than hunger-driven feeding alone [14].
Additional research indicates that oxytocin may also influence metabolic signaling pathways linked to glucose regulation and insulin sensitivity. Some experimental studies have observed modest improvements in postprandial glucose handling and reductions in reward-driven food seeking after oxytocin administration. These findings support the idea that oxytocin may act as a neuromodulator connecting satiety signaling, reward processing, and metabolic regulation, particularly in environments where highly palatable foods can override normal appetite control [13,14].
Appetite regulation represents an important physiological target. Dysregulated feeding behavior can contribute to obesity, insulin resistance, metabolic syndrome, and cardiovascular disease. By influencing hypothalamic appetite signaling and reducing reward-driven eating behaviors, oxytocin may help support healthier energy balance and metabolic resilience over time, particularly when combined with lifestyle strategies that support long-term metabolic health.
Human feeding studies provide measurable evidence for these effects. In controlled laboratory experiments, intranasal oxytocin administration has been shown to reduce caloric intake during test meals by approximately 10–15% (roughly 70-100 kcal for an average meal around 700 total kcal), with particularly pronounced reductions in high-sugar snack consumption and reward-driven eating
Bone Metabolism and Skeletal Health
Beyond its effects on the brain and metabolic signaling, oxytocin may also influence skeletal biology and bone remodeling. Oxytocin receptors are expressed on both osteoblasts, the cells responsible for bone formation, and osteoclasts, which regulate bone resorption. This receptor distribution suggests that oxytocin participates in the regulatory balance between bone building and bone breakdown that maintains skeletal integrity throughout life [15].
Experimental and mechanistic studies provide compelling early evidence for this role. Oxytocin signaling has been shown to increase osteoblast differentiation and bone formation activity by roughly 30–40% in experimental models, while also influencing osteoclast activity in ways that support balanced bone remodeling [16]. In animal models of estrogen-deficiency osteoporosis, oxytocin administration has been shown to restore bone microarchitecture and significantly improve bone mineral density compared with untreated controls, suggesting that oxytocin signaling may help counteract some of the skeletal changes associated with hormonal aging.
More recent reviews of oxytocin biology highlight the hormone’s potential relevance to age-related bone loss, particularly in postmenopausal women, where declines in estrogen and other hormonal shifts contribute to reduced bone density [2,17]. Observational research has also reported that lower circulating oxytocin levels are associated with reduced bone mineral density and increased fracture risk, supporting the hypothesis that oxytocin may function as part of a broader endocrine network regulating skeletal metabolism [2,17].
Skeletal health remains a primary determinant of long-term independence and functional longevity. Bone density and structural integrity influence mobility, fall risk, and fracture susceptibility later in life. Although clinical data are still emerging, the growing body of preclinical and observational research suggests that oxytocin may represent an underappreciated hormonal signal involved in maintaining skeletal resilience across the lifespan.
Pain Perception
Oxytocin has also been investigated for its ability to modulate pain perception and nociceptive signaling. Pain is shaped not only by peripheral injury signals but also by emotional state and central nervous system interpretation. Oxytocin interacts with neural circuits in the hypothalamus, amygdala, and spinal cord that regulate both the sensory and affective dimensions of pain [18].
Controlled human studies provide measurable support for this mechanism. In a double-blind crossover trial using laser-evoked painful stimuli, intranasal oxytocin produced reductions in perceived pain intensity of roughly 15–25% compared with placebo, alongside decreases in early cortical evoked potentials linked to nociceptive processing [19]. These findings suggest oxytocin may influence early stages of pain signal transmission within the central nervous system.
Neuroimaging studies further suggest that oxytocin primarily affects the emotional interpretation of pain, modifying how emotional context alters pain unpleasantness rather than acting as a traditional analgesic [20]. Reviews of oxytocin biology also describe potential mechanisms, including modulation of spinal pain pathways, reduced inflammatory signaling, and improved autonomic regulation [21]. Human clinical trials are ongoing, and nearing completion, evaluating intranasal oxytocin in chronic pain conditions (NCT04903002).
Oxytocin: Mode of Delivery
Understanding how oxytocin works in the body requires more than examining its biological effects alone. Mode of delivery can meaningfully influence where oxytocin acts, how quickly it reaches target tissues, and how long its effects persist. Because oxytocin is a peptide hormone with a relatively short half-life in circulation, the route of administration plays an important role in determining both pharmacokinetics and potential clinical applications [24-28].
Researchers and clinicians have therefore explored multiple delivery approaches, including intranasal, sublingual/oral, and transdermal formulations. Each delivery method has advantages and limitations related to absorption, central nervous system exposure, dosing precision, and duration of effect. The sections that follow examine these delivery strategies in greater detail, beginning with the most studied approach in human research, intranasal oxytocin delivery.
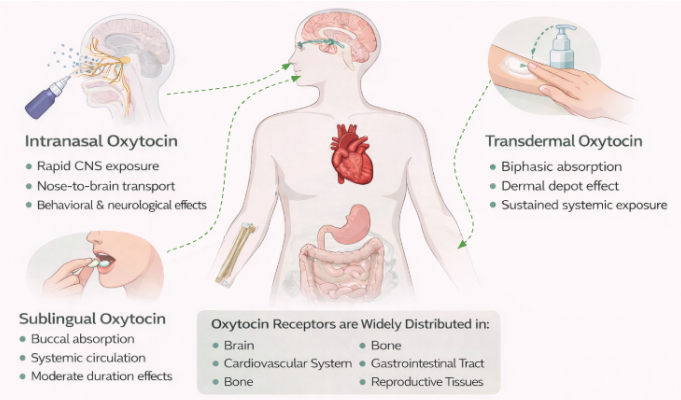
Figure 1. Oxytocin Modes of Delivery and Diverse Biological Effects
Intranasal Oxytocin: Central Delivery and Rapid Pharmacokinetics
Intranasal administration is the most widely studied delivery method for exogenous oxytocin and has become the standard approach in experimental human research. This route is thought to allow oxytocin to access the central nervous system through olfactory and trigeminal nerve pathways, partially bypassing the blood–brain-barrier and enabling more direct engagement with neural circuits involved in stress regulation, emotional processing, and social cognition [24]. Because many of oxytocin’s most studied effects occur within the brain, intranasal delivery has been the preferred method in research examining mood, social behavior, autism spectrum conditions, and stress physiology [25].
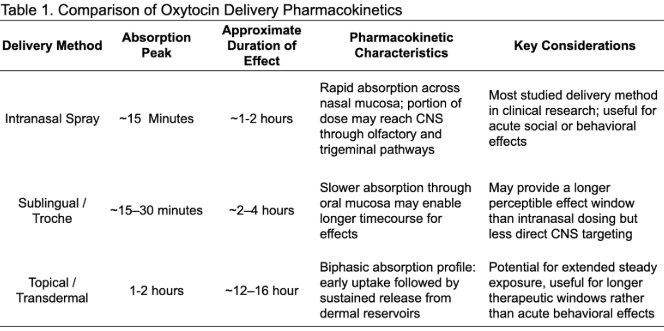
Pharmacokinetically, intranasal oxytocin is characterized by rapid absorption and a relatively short half-life. Pharmacologically relevant plasma or cerebral spinal fluid levels typically appear within 15–30 minutes after administration, and many experimental protocols target behavioral or cognitive tasks 30–60 minutes post-dose, when central nervous system exposure is expected to peak [24-26]. Studies measuring oxytocin dynamics in cerebrospinal fluid suggest a half-life that can be as short as approximately 19 minutes, indicating relatively rapid clearance from central compartments [24]. The functional window of central effects generally lasts about 1-2 hours per dose, during which neural circuits involved in social salience, stress regulation, and emotional processing may be influenced.
Another important consideration is bioavailability variability. Although intranasal delivery may provide a pathway to the brain, systemic absorption into circulation is variable between individuals, which may partly explain why clinical outcomes in intranasal oxytocin studies or personal outcomes can be mixed [26]. Importantly, downstream neural and behavioral effects such as changes in learning, emotional processing, or social cue interpretation may persist beyond the immediate pharmacokinetic window, reflecting oxytocin’s ability to influence neural plasticity and social salience networks even after measurable levels decline.
Sublingual Oxytocin (Troche): Systemic Absorption with Gradual Exposure
Sublingual oxytocin, commonly delivered as a troche, represents another potential route for administering this hormone. In this format, the compound dissolves in the mouth and is absorbed through oral mucosal tissues, allowing oxytocin to enter systemic circulation while bypassing gastrointestinal degradation. Early pharmacokinetic studies demonstrated that oxytocin delivered through oral mucosa can produce measurable systemic effects, although absorption efficiency is variable [27,28]. Some research studies have examined oral oxytocin pharmacokinetics in adult males [27], evaluated a buccal patch delivery system [28], or even “oxipops,” a lollipop delivery [29], highlighting the variety across limited human research for oral mucosal oxytocin delivery.
A key advantage of sublingual delivery is more gradual systemic exposure compared with intranasal dosing. Rather than attempting direct central nervous system delivery through nasal–neural pathways, mucosal administration relies primarily on circulating oxytocin interacting with peripheral receptors and downstream neuroendocrine signaling pathways. Recent research suggests that oral or mucosal oxytocin exposure may still influence behavioral and physiological responses through systemic signaling and possible gut–brain communication mechanisms [29].
Because absorption through oral mucosa occurs gradually, pharmacokinetic effects tend to unfold more slowly than intranasal dosing. Clinical observations often describe onset within approximately 30 minutes, with physiologic effects occurring over the following 2–4 hour window. This profile reflects slower mucosal absorption combined with oxytocin’s rapid systemic clearance, resulting in a longer but less intense exposure curve than intranasal delivery.
Growing interest in peripheral oxytocin signaling pathways and improved formulation strategies has renewed attention to troche-based delivery as a practical, more convenient, flavorful option for longer-duration systemic effects.
Topical Oxytocin: Peripheral Signaling and Sustained Delivery
Topical or transdermal oxytocin, typically delivered as a cream applied to the skin, represents a less studied but emerging route of administration. In this approach, oxytocin is absorbed through the skin and enters systemic circulation, where it can interact with peripheral oxytocin receptors distributed throughout tissues, including the cardiovascular system, reproductive organs, bone, and immune cells [30]. Because the skin acts as a diffusion barrier, transdermal delivery may produce slower and more sustained systemic exposure compared with intranasal administration.
Although peer-reviewed pharmacokinetic studies remain limited, available modeling and clinical observations suggest that topical oxytocin absorption follows a biphasic absorption pattern. Initial uptake may occur within the first 1–2 hours after application, followed by sustained release from dermal reservoirs, which is a significant advantage that may be associated with transdermal (topical) oxytocin [30].
Dermal reservoir release observed with other compounds using similar transdermal bases can produce a prolonged pharmacologic window of approximately 12–16 hours, supporting extended peripheral receptor signaling throughout the day or overnight [30]. Additional discussion of these pharmacokinetic characteristics and formulation strategies is described in technical whitepaper analyses of topical oxytocin delivery [30].
The primary limitation of this route is the lack of robust clinical research, particularly regarding whether meaningful concentrations reach central nervous system targets. Transdermal oxytocin acts primarily through peripheral receptor pathways in the skin, which may influence processes such as metabolic signaling, tissue repair, and autonomic regulation. As research evolves, topical delivery may be best understood as a strategy for longer-duration systemic signaling, complementing faster-acting approaches such as intranasal administration that more directly target central oxytocin pathways but exit the system more rapidly.
Mode of Delivery Should Match Targeted Effects
Because oxytocin is a large peptide hormone with a relatively short half-life, the method of delivery plays a central role in determining how it behaves in the body and how individuals experience its effects. Intranasal administration is currently the most studied approach and is often used when the goal is to influence central nervous system pathways related to stress regulation, social cognition, and emotional processing. By contrast, sublingual or troche formulations may provide more gradual systemic exposure, while transdermal approaches may support long-duration peripheral signaling.
Importantly, clinical responses to oxytocin can vary substantially between individuals. Differences in nasal absorption efficiency, receptor sensitivity, metabolic clearance, and the relative importance of central versus peripheral signaling may all influence how a person responds to a given formulation. For this reason, the route of administration should not be viewed as a purely technical detail, but rather as a meaningful component of oxytocin physiology that may shape how the hormone is best used to support stress regulation, mood, sleep, metabolic balance, and other healthspan-related outcomes.
Personalizing Oxytocin for Optimal Outcomes
Like many signaling molecules in human physiology, oxytocin responses vary widely between individuals. Differences in oxytocin receptor expression, stress history, attachment patterns, baseline autonomic tone, and overall metabolic health can all influence how a person responds to exogenous oxytocin. These factors shape how effectively oxytocin signaling integrates into broader neuroendocrine and autonomic systems that regulate mood, stress recovery, sleep, and social behavior. For this reason, oxytocin is often best approached as a personalized intervention rather than a one-size-fits-all therapy, ideally guided by clinicians who understand both its physiology and the broader context of an individual’s health.
In practice, personalization may involve refining delivery methods and broader regulatory strategies. Some individuals respond best to intranasal delivery, while others may experience more consistent benefits from oral mucosal or topical formulations. In certain cases, simply adjusting the mode of delivery can improve outcomes when early responses appear limited. Exploration of supportive strategies that complement oxytocin, including sleep optimization, stress management interventions, metabolic health improvement, or therapies such as low-dose naltrexone, can augment oxytocin. Strengthening neuroimmune, inflammatory, and autonomic balance may help enhance overall responsiveness within the oxytocin signaling network.
Sex Differences in Oxytocin Signaling
Emerging research suggests that oxytocin’s physiological effects may differ somewhat between males and females, largely due to interactions with sex hormones and differences in receptor expression patterns. Oxytocin appears to follow estrogen levels closely [22]. Estrogen may increase oxytocin receptor expression and signaling sensitivity, especially in brain regions involved in emotional regulation, social cognition, and stress processing [23]. This interaction may help explain why several studies report stronger oxytocin-related effects in domains such as stress buffering, sleep regulation, and bone metabolism in female populations, particularly during life stages influenced by reproductive hormones, including the menstrual cycle, pregnancy, and menopause [2,22,23].
Across the lifespan, oxytocin signaling appears to interact dynamically with sex hormones. Estrogen tends to amplify oxytocin receptor activity, while testosterone and other androgens may shape oxytocin-related social and behavioral responses differently in males [23]. These interactions help coordinate biological processes ranging from maternal caregiving and pair bonding to sexual behavior and parental investment. At the same time, oxytocin also contributes to male physiology through effects on emotional regulation, social bonding, and paternal behavior [9,10,22].
Rather than indicating that oxytocin is inherently more important for one sex than the other, current evidence suggests that the contexts and magnitude of oxytocin signaling differ between males and females across different life stages [22,23]. Recent research also highlights that oxytocin may interact with sex hormones to shape neural plasticity, stress resilience, and social learning across the lifespan, suggesting that oxytocin signaling adapts alongside broader endocrine changes over time [23]. These findings are beginning to inform more personalized approaches to oxytocin-based therapies that account for sex, hormonal status, and life stage; it may partially explain why oxytocin can be particularly beneficial in women especially around midlife and menopause transition [2] when endogenous oxytocin levels are in rapid decline or find a lower baseline between the ages of 40 and 65 years (Figure 2) [22,23].
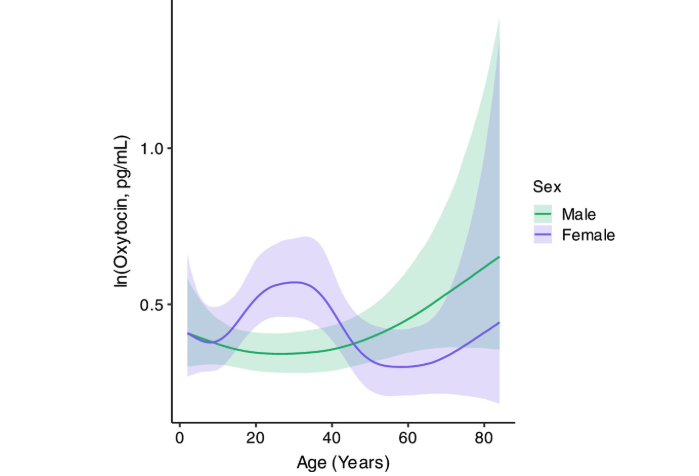
Figure 2. Oxytocin concentration across the lifespan
Dosing Frequency and Maintaining Receptor Sensitivity
A common clinical question is why oxytocin is not typically recommended for continuous daily use without breaks. Like many neuropeptide signaling systems, oxytocin receptors can undergo downregulation or desensitization when exposed to persistent stimulation. When receptor systems are overstimulated for prolonged periods, the body may temporarily reduce receptor sensitivity as a way to maintain physiologic balance. This phenomenon has been observed across multiple hormone and neurotransmitter systems and is one reason dosing strategies for signaling molecules are often designed with periodic pauses.
In practice, many clinicians recommend intermittent dosing patterns to help preserve receptor responsiveness. A common approach is maintaining two consecutive days off per week, allowing oxytocin receptors and downstream signaling pathways time to reset. While formal clinical trials on optimal oxytocin dosing schedules remain limited, this strategy reflects a broader principle in neuroendocrine physiology: maintaining sensitivity in signaling systems often requires periods of recovery between stimulation cycles.
Lifestyle Foundations That Amplify Oxytocin Signaling
Pharmacologic oxytocin is best viewed as a supportive amplifier of healthy physiology, not a replacement for foundational lifestyle practices that naturally regulate the oxytocin system. Endogenous oxytocin signaling is closely tied to the body’s autonomic balance, emotional regulation, and metabolic health. Behaviors that improve these systems may therefore enhance the body’s responsiveness to both endogenous and exogenous oxytocin.
Several lifestyle factors appear particularly supportive of oxytocin signaling. Resistance training, high-quality sleep, meaningful social interaction, metabolic health optimization, and effective stress management all influence neuroendocrine regulation and autonomic balance [1,2]. These habits strengthen the physiologic environment in which oxytocin operates, potentially improving therapeutic response while also supporting broader healthspan goals. When pharmacologic therapy and lifestyle foundations align, the oxytocin system may function more effectively as part of the body’s integrated network for resilience, recovery, and social well-being.
Assessing Oxytocin Response: Biomarkers and Physiologic Signals
One challenge with oxytocin therapy is that its benefits can be subtle, multi-system, and sometimes gradual. Because oxytocin influences autonomic regulation, emotional processing, metabolism, and social signaling simultaneously, improvements may appear across several physiologic domains rather than as a single dramatic effect. For this reason, evaluating response often requires looking beyond one symptom and observing a broader set of physiologic and behavioral signals over time [31].
Importantly, some benefits may be developing even if the initial goal, such as stress relief, does not immediately feel dramatic. Clinical researchers often assess oxytocin-related effects using a combination of objective biomarkers and subjective experience markers. A 2025 publication in the Proceedings of the National Academy of Sciences of the United States of America reports there is concentration-dependent higher self-reported health associated with higher oxytocin levels for both women and men (Figure 3) [23]. Those who have higher oxytocin levels also subjectively report better health. These signals can help individuals and clinicians refine dosing strategies, adjust delivery methods, or simply recognize that meaningful physiologic changes are occurring even when subtle or related to subjective perceived health or linked with specific experiences.
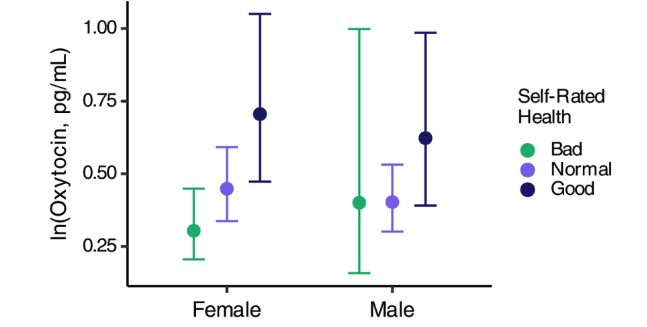
Figure 3. Oxytocin Concentration Association with Self-Rated Health
Physiologic and Wearable-Based Signals
Wearable technology can provide useful insights into changes in autonomic balance and recovery physiology. Metrics to monitor may include:
- Heart Rate Variability (HRV) – Higher HRV often reflects improved parasympathetic nervous system tone and better resilience to stress.
- Resting Heart Rate – A gradual reduction may indicate improved autonomic balance and cardiovascular recovery capacity.
- Sleep metrics – Devices may reveal improvements in:
- sleep duration
- sleep efficiency
- deep sleep stages
- reduced nighttime awakenings or fragmentation
These physiologic signals can sometimes shift before subjective mood changes are fully noticeable.
Cardiometabolic and Stress-Related Signals
Oxytocin also interacts with systems involved in metabolic regulation and cardiovascular health. Potential changes that may emerge over time include:
- Reduced resting blood pressure
- Improved stress recovery scores on wearable platforms
- More stable daytime energy and mood rhythms
- Subtle improvements in appetite regulation or eating behavior
While these signals are not exclusive to oxytocin, improvements across several domains may indicate that autonomic and neuroendocrine balance is shifting.
Subjective Experience Markers
Subjective observations remain an important part of evaluating oxytocin response. Individuals may notice:
- Greater emotional regulation during stressful situations
- Reduced stress reactivity or rumination
- Improved sleep onset or nighttime calm
- A stronger sense of social connection or empathy
- Changes in pain perception or recovery after exertion
- More stable appetite signals or satiety awareness
It is also important to recognize that oxytocin’s multi-system effects may not align perfectly with the initial reason someone begins therapy. For example, an individual seeking stress relief may instead notice improved sleep quality, appetite control, or social engagement first.
For these reasons, evaluating oxytocin therapy is best approached as an ongoing feedback process rather than a single yes-or-no outcome. By tracking physiologic signals, wearable data, and subjective experience over several weeks, individuals and clinicians can make informed adjustments to dosing schedules, delivery methods, or supporting lifestyle practices before deciding whether the intervention is beneficial.
Oxytocin for Restoration of Physiologic Balance
Oxytocin is increasingly recognized as a multisystem regulatory hormone that influences far more than social bonding alone. Research suggests it interacts with systems involved in stress regulation, emotional processing, sleep quality, appetite signaling, metabolic function, pain perception, and skeletal health. Because these systems are deeply interconnected, oxytocin’s effects often appear as subtle improvements across several areas of physiology rather than a single dramatic change.
When used thoughtfully under appropriate clinical guidance and combined with foundational habits such as quality sleep, resistance training, stress management, and metabolic health optimization, oxytocin may help restore regulatory balance across neuroendocrine and autonomic systems. In this sense, oxytocin is not a performance-enhancing shortcut, but a signal that may support greater resilience, recovery, and long-term healthspan as part of an integrated approach to well-being.
- Quintana, D.S., Lischke, A., Grace, S. et al. Advances in the field of intranasal oxytocin research: lessons learned and future directions for clinical research. Mol Psychiatry 26, 80–91 (2021). https://doi.org/10.1038/s41380-020-00864-7
- Dunietz, G. L., Tittle, L. J., Mumford, S. L., O'Brien, L. M., Baylin, A., Schisterman, E. F., Chervin, R. D., & Young, L. J. (2024). Oxytocin and women's health in midlife. The Journal of endocrinology, 262(1), e230396. https://doi.org/10.1530/JOE-23-0396
- Gutkowska, J., & Jankowski, M. (2008). Oxytocin revisited: It is also a cardiovascular hormone. Journal of the American Society of Hypertension : JASH, 2(5), 318–325. https://doi.org/10.1016/j.jash.2008.04.004
- Breuil, V., Fontas, E., Chapurlat, R., Panaia-Ferrari, P., Yahia, H. B., Faure, S., Euller-Ziegler, L., Amri, E. Z., & Szulc, P. (2015). Oxytocin and bone status in men: analysis of the MINOS cohort. Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA, 26(12), 2877–2882. https://doi.org/10.1007/s00198-015-3201-3
- Jurek, B., & Neumann, I. D. (2018). The Oxytocin Receptor: From Intracellular Signaling to Behavior. Physiological reviews, 98(3), 1805–1908. https://doi.org/10.1152/physrev.00031.2017
- Cardoso, C., Kingdon, D., & Ellenbogen, M. A. (2014). A meta-analytic review of the impact of intranasal oxytocin administration on cortisol concentrations during laboratory tasks: moderation by method and mental health. Psychoneuroendocrinology, 49, 161–170. https://doi.org/10.1016/j.psyneuen.2014.07.014
- Young Kuchenbecker, S., Pressman, S. D., Celniker, J., Grewen, K. M., Sumida, K. D., Jonathan, N., Everett, B., & Slavich, G. M. (2021). Oxytocin, cortisol, and cognitive control during acute and naturalistic stress. Stress (Amsterdam, Netherlands), 24(4), 370–383. https://doi.org/10.1080/10253890.2021.1876658
- Chaulagain, R. P., Shrestha, Y., Shrestha, H., Bhandari, R., & Gurung, P. (2025). The neurobiological impact of oxytocin in mental health disorders: a comprehensive review. Annals of medicine and surgery (2012), 87(3), 1479–1486. https://doi.org/10.1097/MS9.0000000000003015
- Triana-Del Rio, R., Ranade, S., Guardado, J., LeDoux, J., Klann, E., & Shrestha, P. (2022). The modulation of emotional and social behaviors by oxytocin signaling in limbic network. Frontiers in molecular neuroscience, 15, 1002846. https://doi.org/10.3389/fnmol.2022.1002846
- Matsushita, H., & Nishiki, T. I. (2025). Human social behavior and oxytocin: Molecular and neuronal mechanisms. Neuroscience, 570, 48-54. https://pubmed.ncbi.nlm.nih.gov/39961388/
- Braga, R. I., Panaitescu, A., Bădescu, S., Zăgrean, A. M., & Zăgrean, L. (2014). Intranasal administration of oxytocin alters sleep architecture. Biological Rhythm Research, 45(1), 69-75. https://www.semanticscholar.org/paper/Intranasal-administration-of-oxytocin-alters-sleep-Braga-Panaitescu/4773504965d1c3666b45f5e0658adce3f545e33f
- Liu, Y. C., Deng, Y. C., Zhu, Z. T., Rao, B., Shang, H. L., Wang, L. K., Li, T., Wang, Y. R., Wang, J. Z., Zhang, Q. P., Gao, Y., & Xu, H. B. (2025). Oxytocin modulates inhibitory balance in the prelimbic cortex to support social memory consolidation during REM sleep. Theranostics, 15(8), 3257–3274. https://doi.org/10.7150/thno.109104
- Morton, G. J., Thatcher, B. S., Reidelberger, R. D., Ogimoto, K., Wolden-Hanson, T., Baskin, D. G., Schwartz, M. W., & Blevins, J. E. (2012). Peripheral oxytocin suppresses food intake and causes weight loss in diet-induced obese rats. American journal of physiology. Endocrinology and metabolism, 302(1), E134–E144. https://doi.org/10.1152/ajpendo.00296.2011
- Blevins, J. E., Graham, J. L., Morton, G. J., Bales, K. L., Schwartz, M. W., Baskin, D. G., & Havel, P. J. (2015). Chronic oxytocin administration inhibits food intake, increases energy expenditure, and produces weight loss in fructose-fed obese rhesus monkeys. American journal of physiology. Regulatory, integrative and comparative physiology, 308(5), R431–R438. https://doi.org/10.1152/ajpregu.00441.2014
- Mohammadi, S. M., Karimi, S., Saniee, N., Kamyari, N., & Radmanesh, E. (2024). The Effect of Oxytocin on Osteoporosis Improvement: A Systematic Review. Medical journal of the Islamic Republic of Iran, 38, 140. https://doi.org/10.47176/mjiri.38.140
- Colaianni, G., Sun, L., Zaidi, M., & Zallone, A. (2014). Oxytocin and bone. American journal of physiology. Regulatory, integrative and comparative physiology, 307(8), R970–R977. https://doi.org/10.1152/ajpregu.00040.2014
- Wang T, Ye J, Zhang Y, Li J, Yang T, Wang Y, Jiang X and Yao Q (2024) Role of oxytocin in bone. Front. Endocrinol. 15:1450007. doi: 10.3389/fendo.2024.1450007 https://doi.org/10.3389/fendo.2024.1450007
- Long, P., Scholl, J. L., Wang, X., Kallsen, N. A., Ehli, E. A., & Freeman, H. (2023). Intranasal Oxytocin and Pain Reduction: Testing a Social Cognitive Mediation Model. Brain Sciences, 13(12), 1689. https://doi.org/10.3390/brainsci13121689
- Boll, S., Almeida de Minas, A. C., Raftogianni, A., Herpertz, S. C., & Grinevich, V. (2018). Oxytocin and Pain Perception: From Animal Models to Human Research. Neuroscience, 387, 149–161. https://doi.org/10.1016/j.neuroscience.2017.09.041
- Lopes, S., & Osório, F. L. (2023). Effects of intranasal oxytocin on pain perception among human subjects: A systematic literature review and meta-analysis. Hormones and behavior, 147, 105282. https://doi.org/10.1016/j.yhbeh.2022.105282
- Mekhael, A. A., Bent, J. E., Fawcett, J. M., Campbell, T. S., Aguirre-Camacho, A., Farrell, A., & Rash, J. A. (2023). Evaluating the efficacy of oxytocin for pain management: An updated systematic review and meta-analysis of randomized clinical trials and observational studies. Canadian journal of pain = Revue canadienne de la douleur, 7(1), 2191114. https://doi.org/10.1080/24740527.2023.2191114
- Quintana, D. S., Glaser, B. D., Kang, H., Kildal, E. S., Audunsdottir, K., Sartorius, A. M., & Barth, C. (2024). The interplay of oxytocin and sex hormones. Neuroscience & Biobehavioral Reviews, 163, 105765. https://www.sciencedirect.com/science/article/pii/S0149763424002343?via%3Dihub
- Colby, A. E., Jud, D. C., Baettig, V., Martin, J. S., Scaff, C., Gurven, M. D., Trumble, B. C., Beheim, B. A., Hooper, P. L., Cummings, D. K., Kaplan, H., Stieglitz, J., Ista, A. C., & Jaeggi, A. V. (2025). Oxytocin varies across the life course in a sex-specific way in a human subsistence population. Proceedings of the National Academy of Sciences of the United States of America, 122(51), e2509977122. https://doi.org/10.1073/pnas.2509977122
- Quintana, D.S., Lischke, A., Grace, S. et al. Advances in the field of intranasal oxytocin research: lessons learned and future directions for clinical research. Mol Psychiatry 26, 80–91 (2021). https://doi.org/10.1038/s41380-020-00864-7
- Martins, D., Brodmann, K., Veronese, M., Dipasquale, O., Mazibuko, N., Schuschnig, U., ... & Paloyelis, Y. (2022). Less is more. Progress in Neurobiology, 211. https://www.sciencedirect.com/science/article/pii/S0301008222000259
- Leake, R. D., Weitzman, R. E., & Fisher, D. A. (1980). Pharmacokinetics of oxytocin in the human subject. Obstetrics and gynecology, 56(6), 701–704. https://pubmed.ncbi.nlm.nih.gov/7443113/
- De Groot, A. N., Vree, T. B., Hekster, Y. A., Pesman, G. J., Sweep, F. C., Van Dongen, P. J., & Van Roosmalen, J. (1995). Bioavailability and pharmacokinetics of sublingual oxytocin in male volunteers. The Journal of pharmacy and pharmacology, 47(7), 571–575. https://doi.org/10.1111/j.2042-7158.1995.tb06716.x
- Li, C., Bhatt, P. P., & Johnston, T. P. (1996). In vitro release and permeation of oxytocin from a mucoadhesive buccal patch. Pharmaceutical development and technology, 1(4), 357–364. https://doi.org/10.3109/10837459609031430
- Xu, D., Lan, C., Kou, J., Yao, S., Zhao, W., & Kendrick, K. M. (2024). Oromucosal Administration of Oxytocin: The Development of 'Oxipops'. Pharmaceutics, 16(3), 333. https://doi.org/10.3390/pharmaceutics16030333
- Gabriel, A., Sepah, S.C., Krentz, S.A Novel Transdermal Oxytocin Formulation Delivers Sustained Mood, Stress, and Sleep Benefits. Whitepaper, Accessed March, 2026.
- DuBois, M., Tseng, A., Francis, S. M., Haynos, A. F., Peterson, C. B., & Jacob, S. (2022). Utility of Downstream Biomarkers to Assess and Optimize Intranasal Delivery of Oxytocin. Pharmaceutics, 14(6), 1178. https://doi.org/10.3390/pharmaceutics14061178
Related studies