Semaglutide is an incredibly powerful weight loss tool in individuals with Type II Diabetes & Obesity, resulting in up to 15kg of weight loss.
The primary mechanism of its weight loss effect is due to changes in brain signaling related to satiety, appetite suppression & prolonging the gastric emptying following a meal.
The weight loss effect is a combination of fat mass and lean muscle mass due to severe and chronic low energy intake.
Preventing muscle loss with Semaglutide should focus on resistance exercise training and combination with sufficient dietary protein to mitigate the loss of muscle.
The world of GLP-1 agonists is a rapidly growing field, with manufacturers now attempting to utilize dual (Mazdutide & tirzepatide) and even tri-agonist (LY3437943) formulations to target multiple mechanisms and further accelerate weight loss.
Overview:
Since GLP-1 was first discovered in the 1970s, and its subsequent role in insulin regulation was identified in 1987, its role in human physiology remained largely unexplored for decades. However, in early 2005, the development of GLP-1 receptor agonists took the world by storm. Following a recent wave of high-impact scientific publications in the New England Journal of Medicine and The Lancet, GLP-1 receptors have shown incredibly promising data from randomized, double-blind, placebo-control trials to drastically reduce body mass and in particular, body fat in patients with obesity & type II diabetes.
However, amidst the ever-growing popularity of these drugs,Dr. Peter Attia, has been vocal on social media, blogs, and podcasts regarding the potential downsides and adverse effects of GLP-1 drugs on muscle mass. Here we provide more depth behind Peter's opinion and dive deeper into the relationship between GLP1 & muscle mass.
First of all, what is GLP-1?
GLP-1 (Glucagon-Like Peptide-1) is a hormone that is essential in regulating blood sugar levels and appetite [1]. It is released by specialized cells in the small intestine in response to a meal [2]. This hormone, among several other gut-derived hormones, acts on various organs in the body, including the pancreas, stomach, and brain, to exert its effects [1]. One of the primary functions of GLP-1 is to stimulate insulin release from the pancreas [3]. Insulin helps lower blood sugar levels by facilitating glucose uptake from the bloodstream into cells. GLP-1 also inhibits the release of glucagon, another pancreatic hormone that raises blood sugar levels. By increasing insulin secretion and reducing glucagon release, GLP-1 helps maintain glucose homeostasis.
Additionally, GLP-1 slows down the emptying of the stomach, which leads to a feeling of fullness and reduces appetite. It also acts on the brain to regulate food intake and promote satiety, thus contributing to weight management. We will dive into how GLP-1 receptor agonists alter these mechanisms later on!
What are GLP-1 receptor agonists?
GLP-1 receptor agonists are a type of therapeutic that activates the GLP-1 receptors in various tissues, mimicking the effects of the naturally occurring hormone glucagon-like peptide-1 (GLP-1). Therefore, GLP-1 receptor agonists are primarily used in managing type II diabetes and, in some cases, for weight loss. Interestingly, GLP-1 receptor agonists resist degradation by the enzyme DPP-4 (dipeptidyl peptidase-4), which usually breaks down natural GLP-1 and reduces its activity [4]. This makes GLP-1 receptor agonists more potent than naturally occurring GLP-1 and allows for once-weekly dosing in some cases.
Since their initial development, the market size of GLP-1 receptor drugs has reached an estimated $12.7 billion per year, with an annual growth rate of >6% per year – Overall, making this an incredibly lucrative drug for manufacturers such as Novo Nordisk, Eli Lilly, and AstraZeneca.
What forms of GLP-1 drugs are there?
GLP-1 receptor agonists come in different forms, including injectable formulations (Exenatide, Liraglutide, Dulaglutide, and Semaglutide) and oral formulations (Semaglutide). The benefit of Semaglutide is the significantly longer half-life compared to other forms (160 vs 12 hrs).
They are typically used in combination with other diabetes medications, such as metformin, which lead to drastic weight loss and improved cardiovascular outcomes & metabolic outcomes.
Lately, there has been a recent drive-by pharmaceutical company to develop dual (Mazdutide) and even tri-agonist drugs (LY3437943) that target GLP, GIP (glucose-dependent insulinotropic peptide), and Glucagon receptors, which 'in theory' combine the anorectic and insulinotropic activity of GLP-1 & GIP, with the energy expenditure effects of Glucagon – Overall, tackling weight loss at three distinct angles. These multi-agonists are incredibly new, and we'll touch upon these towards the end of the article. For this article, we will primarily focus on Semaglutide and its effect on human physiology & metabolism.
Popularity & Media Attention?
Since the first GLP-1 receptor agonist drug, Exenatide, was approved by the FDA in 2005, there have been several iterations of these drugs, with the most widely established being, Semaglutide. Notably, Semaglutide has been hailed as a 'wonder drug' and has been subjected to numerous high-profile randomized, double-blind, placebo trials under the following trial names:
- SUSTAIN Trial: The trials on the SUSTAIN series investigate theonce-weekly injectable of Semaglutide. There are currently 15 published articles from this trial, with one currently in the process of recruiting volunteers.
- PIONEER Trial: The trials in the PIONEER series test various aspects of the oral version of Semaglutide. There are currently 10 published articles on this trial, with 6 more currently unpublished or in the process of recruiting volunteers.
- STEP Trial: The trial investigates the effects of a higher dose of one-weekly injectable Semaglutide. In particular, compared to the approved 1.0mg dose, researchers are investigating doses from 2.4mg up to 7.2mg per week. There are currently 7 published articles from this trial, with a further 8 either unpublished or recruiting volunteers.
- OASIS Trial: The trial investigates the effects of a daily oral Semaglutide dose of 50mg in individuals living with obesity. Data from this trial have recently become available as of 22nd May.
As with most things, the popularity of weight-loss drugs has skyrocketed, with even celebrities endorsing their use. In particular, the media have reported multi-billionaires Elon Musk, Michael Rubin, and several other high-profile celebrities using Semaglutide in an attempt to facilitate weight loss.
Alarmingly, and we agree with Dr Peter Attia's recent statement on his blog regarding the use of Semaglutide in non-diseased and apparently healthy individuals – As these drugs have only been tested on individuals that are obese, we currently don't know how these drugs affect 'normal weight' and 'healthy' individuals.
Mechanisms of Semaglutide in Human Health & Disease Prevention
Semaglutide exerts its effects through several physiological and biochemical mechanisms. Notably, when administered via weekly subcutaneous injection or a daily oral dose, Semaglutide binds to GLP-1 receptors, activating a cascade of intracellular signaling pathways [1]. Overall, this leads to increased insulin secretion from pancreatic beta cells, which helps regulate blood sugar levels [5]. Semaglutide also inhibits glucagon secretion, reducing hepatic glucose production and improving glycaemic control [6]. However, an effective mechanism is its ability to alter brain signaling [7], slow gastric emptying [8], enhancing satiety and reducing food intake [9]. These mechanisms collectively improve glucose control, reduce appetite, and promote significant and clinically meaningful weight loss. Here we provide a brief overview of the latter mechanisms related to the molecular control of appetite, as these are likely the key drivers in the drastic weight loss seen with this drug.
Appetite, Hunger & Food Intake:
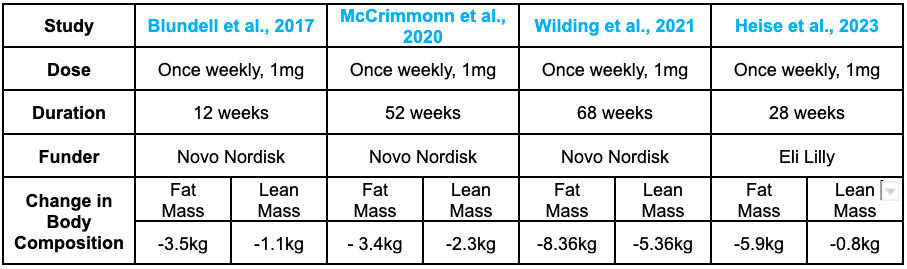
A 2017 study completed By Professor John Blundell at the University of Leeds in collaboration with Novo Nordisk showed that 12 weeks of a once-weekly dose of Semaglutide led to a ~300kcal lower energy intake following an ad libitum (i.e., an unrestricted breakfast) [10]. This translated into a ~24% decrease in energy intake across an entire day. Over the 12-week intervention, there was also a ~5kg reduction in body mass, in which 3.5kg was fat, with a 1.1kg loss in lean mass. The mechanism behind this phenomenon was recently unraveled by Professor Lotte Knudsens in 2020 – Unsurprisingly, this was due to Semaglutide's effect on the brain. As the chief scientific advisor of the Global Drug Discovery Group at Novo Nordisk, her team observed, in mice, a 22% reduction in body weight, coupled with a ~17% reduction in food intake and an increase in daily energy expenditure during the day [7].
To find out the mechanisms behind why Semaglutide has such a potent effect, they injected a florescent version of the drug to determine its uptake into the brain. They observed a significant increase in Semaglutide in the brain stem with considerable accumulation within the septal nucleus and hypothalamus, but interestingly did not cross the blood-brain barrier [7].
Unlike human studies, where sampling tissues and biofluids are primarily limited to blood plasma, muscle, adipose, urine & fecal samples, mouse models allow researchers to sample multiple organs and tissues, often impossible due to ethical limitations in human volunteers. In Professor Knudsen's paper, they harvested the brain at the end of the study and analyzed the brain tissue for specific genes that may regulate this change in brain function. Through RNA-sequencing technology, they identified Semaglutide increased the expression of Prolactin Releasing Peptide [7]. This peptide is released by neurons in the hypothalamus, which reduces food intake [11].
Among the molecular signaling in the brain, Semaglutide also has powerful effects on delaying gastric emptying and prolonging the feeling of fullness [8].
Notably, the GLP-1-mediated mechanism is due to a relaxation in stomach muscles. In a 12-week randomized, placebo control trial, Semaglutide was observed to slow the gastric emptying following a meal by ~30% 4hrs following ingestion. This resulted in 37% of the solid meal being retained in the stomach at the end of the 4-hour post-meal period, compared to no remaining meal in the placebo group [8].
Therefore, in concert with the increases in insulin secretion, and peripheral insulin sensitivity, Semaglutide's potential primary mechanism may be altering brain function and satiety, thereby reducing calorie intake in patient populations.
GLP-1 RA's on Body Mass & Lean Mass:
A seminal study published in the New England Journal of Medicine in 2021 by Professor John Wilding, which has already received >1,000 citations, showed a remarkable reduction in body mass with a once-weekly Semaglutide dose of 2.4mg for 68 weeks [12].
This study, for the first time, showed Semaglutide to reduce body mass by 14.85kg, albeit with 44% & 31% of participants reporting side effects of nausea or diarrhea [12]. Similar longer-term studies as part of the STEP Trail published in Nature Medicine showed up to 2 years of Semaglutide treatment induced similar weight loss of ~16kg, showing a tapering off in weight loss, where no further weight loss is achieved [13].
However, although Semaglutide resulted in large weight loss, these studies are unable to determine how much of this weight loss was from fat. As there are now several published articles on Semaglutide on weight, we are aiming to focus our attention on the effects of this drug on body composition, so only studies that have measured body composition via DEXA are of interest to us – This way, we can separate the effects of these drugs on fat mass and lean muscle mass.
Effects of GLP-1 Agonists on Fat Mass & Lean Mass:
Initial body composition data from a short-term (12-week) Semaglutide treatment observed a 5kg change in body mass, with 70% (3.5kg) of that from fat mass and 22% (1.1kg) from lean mass [14].
2020 data from Novo Nordisk's SUSTAIN trial showed an impressive reduction in total body mass of 5.7kg, in which 3.4kg (60%) was fat-mass and 2.3kg (60%) loss in lean muscle mass following 52 weeks of Semaglutide at a dose of 1mg, once per week [15]. This was followed up with a STEP trial from Professor John Wilding. In this study, as mentioned above, they observed a 15.3kg reduction in body weight, with 8.36kg (58%) from fat mass and 5.26kg (34.3%) from lean muscle mass 12. More recent clinical trials have observed a large reduction of 6.9kg in total body mass, with 5.9kg (85%) from fat mass and only 0.8kg (11%) from lean mass [16].
Why the Declines in Lean Mass?
So, based on the data above, there has been a lot of talk on social media exaggerating the effects of Semaglutide on declines in lean mass. Here we attempt to provide an explanation as to why this occurs.
Firstly, Semaglutide causes a considerable reduction in lean mass in clinical trials. However, previous evidence has suggested they do, in fact, have a positive effect on muscle protein synthesis. Notably, the long-term treatment with inhibitors of the GLP‐1‐degrading enzyme, dipeptidyl peptidase‐4 (DPP‐4), demonstrates improved muscle mass preservation in older age [17].
Recent evidence published in Aging Cell by Professor Philip Atherton's group at the University of Nottingham showed the infusion of GLP1 increased the post-meal protein synthetic response by ~62%, while the placebo only increased by ~29% [18]. Although the precise mechanism is unclear, the authors suggest it could be due to the increased microvascular blood flow observed in their study [18].
So where is lean mass loss coming from, and how can it be prevented?
In most, if not all, of the studies, they are purely looking at the effect of Semaglutide independent from exercise. Therefore, the simple explanation as to why individuals in these studies lose so much lean mass is due to (1) the lack of exercise (in particular resistance exercise) and (2) the chronic negative energy balance.
In the few studies that have measured energy intake, they have reported Semaglutide groups consuming 750 kcals (25%) less than the placebo group per day [14].
How bad is energy balance for muscle mass regulation? Evidence from the world-leading Dr. Stefan Pasiakos has shown as little as 10 days of a 20% reduction in energy intake results in a 19% reduction in muscle protein synthesis 19. Interestingly, this study maintained a relatively high protein diet of 1.5kg/per day and still lost ~1kg of body weight in 10 days [19]. This clearly shows the importance of exercise, particularly resistance exercise, to maintain muscle mass when in a negative energy balance.
Evidence of this has been reported from the prestigious lab of Professor Stuart Phillips at McMaster University, Canada. Research conducted by an MSc student at the time, Thomas Longland, showed that completing regular resistance exercise training in a severe energy deficit of 40% for 4 weeks prevented any loss in lean muscle mass [20]. The study reported a 3.5kg loss in fat mass, with no detectable changes in lean muscle mass [20].
Future research should clearly seek methods to co-administer Semaglutide with a structured resistance exercise training program to maintain as much muscle as possible during a weight-loss journey. However, instead of putting in the hard work, time, and effort into exercise, there are now clinical trials underway in collaborating with Versanis Biotech to pair Semaglutide with bimagrumab (a highly potent drug known for preventing muscle wasting via the activin type II receptor). However, these are still underway, and we look forward to seeing these data in the next year or two.
- Drucker DJ. Cell Metabolism Review Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metab 2018; 27: 740–756.
- Baggio LL, Drucker DJ. Biology of Incretins: GLP-1 and GIP. 2007. doi:10.1053/j.gastro.2007.03.054.
- Drucker DJ, Philippe J, Mojsov S, Chick WL, Habener JF. Glucagon-like peptide I stimulates insulin gene expression and increases cyclic AMP levels in a rat islet cell line. Proceedings of the National Academy of Sciences 1987; 84: 3434–3438.
- Lau J, Bloch P, Schä L, Pettersson I, Spetzler J, Kofoed J et al. Discovery of the once-weekly glucagon-like peptide-1 (GLP-1) analogue semaglutide. ACS Publications 2015; 58: 7370–7380.
- Kapitza C, Dahl K, Jacobsen JB, Axelsen MB, Flint A. Effects of semaglutide on beta cell function and glycaemic control in participants with type 2 diabetes: a randomised, double-blind, placebo-controlled trial. doi:10.1007/s00125-017-4289-0.
- Weghuber D, Barrett T, Barrientos-Pérez M, Gies I, Hesse D, Jeppesen OK et al. Once-Weekly Semaglutide in Adolescents with Obesity. N Engl J Med 2022; 387: 2245.
- Gabery S, Salinas CG, Paulsen SJ, Ahnfelt-Rønne J, Alanentalo T, Baquero AF et al. Semaglutide lowers body weight in rodents via distributed neural pathways. 2020. doi:10.1172/jci.insight.133429.
- Jensterle M, Ferjan S, Ležaič L, Sočan A, Goričar K, Zaletel K et al. Semaglutide delays 4-hour gastric emptying in women with polycystic ovary syndrome and obesity. Diabetes Obes Metab 2023; 25: 975–984.
- Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine 2021; 384: 989–1002.
- Blundell J, Finlayson G, Axelsen M, Flint A, Gibbons C, Kvist T, Hjerpsted JB. Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes Obes Metab. 2017 Sep;19(9):1242-1251. doi: 10.1111/dom.12932. Epub 2017 May 5. PMID: 28266779; PMCID: PMC5573908.
- Jörgensen SKM, Karnošová A, Mazzaferro S, Rowley O, Chen H-JC, Robbins SJ et al. Title: An analogue of the Prolactin Releasing Peptide reduces obesity and promotes adult neurogenesis Authors. doi:10.1101/2023.02.28.528936.
- Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine 2021; 384: 989–1002.
- Garvey WT, Batterham RL, Bhatta M, Buscemi S, Christensen LN, Frias JP, Jódar E, Kandler K, Rigas G, Wadden TA, Wharton S; STEP 5 Study Group. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022 Oct;28(10):2083-2091. doi: 10.1038/s41591-022-02026-4. Epub 2022 Oct 10. PMID: 36216945; PMCID: PMC9556320.
- Blundell J, Finlayson G, Axelsen M, Flint A, Gibbons C, Kvist T et al. Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes Obes Metab 2017; 19: 1242–1251.
- Mccrimmon RJ, Catarig A-M, Frias JP, Lausvig NL, Le Roux CW, Thielke D et al. Effects of once-weekly semaglutide vs once-daily canagliflozin on body composition in type 2 diabetes: a substudy of the SUSTAIN 8 randomised controlled clinical trial. doi:10.1007/s00125-019-05065-8.
- Heise T, DeVries JH, Urva S, Li J, Pratt EJ, Thomas MK et al. Tirzepatide Reduces Appetite, Energy Intake, and Fat Mass in People With Type 2 Diabetes. Diabetes Care 2023; 46: 998.
- Bouchi R, Fukuda T, Takeuchi T, Nakano Y, Murakami M, Minami I, Izumiyama H, Hashimoto K, Yoshimoto T, Ogawa Y. Dipeptidyl peptidase 4 inhibitors attenuates the decline of skeletal muscle mass in patients with type 2 diabetes. Diabetes Metab Res Rev. 2018 Feb;34(2). doi: 10.1002/dmrr.2957. Epub 2017 Nov 23. PMID: 29054111.
- Abdulla H, Phillips BE, Wilkinson DJ, Limb M, Jandova T, Bass JJ, Rankin D, Cegielski J, Sayda M, Crossland H, Williams JP, Smith K, Idris I, Atherton PJ. Glucagon-like peptide 1 infusions overcome anabolic resistance to feeding in older human muscle. Aging Cell. 2020 Sep;19(9):e13202. doi: 10.1111/acel.13202. Epub 2020 Aug 3. PMID: 32744385; PMCID: PMC7511886.
- Pasiakos SM, Vislocky LM, Carbone JW, Altieri N, Konopelski K, Freake HC et al. Acute energy deprivation affects skeletal muscle protein synthesis and associated intracellular signaling proteins in physically active adults. J Nutr 2010; 140: 745–751.
- Longland TM, Oikawa SY, Mitchell CJ, Devries MC, Phillips SM. Higher compared with lower dietary protein during an energy deficit combined with intense exercise promotes greater lean mass gain and fat mass loss: a randomized trial. Am J Clin Nutr. 2016 Mar;103(3):738-46. doi: 10.3945/ajcn.115.119339. Epub 2016 Jan 27. PMID: 26817506.