Muscles are not merely about physical strength; they play a crucial role in maintaining our health and independence, especially as we age. The mechanisms governing muscle growth and shrinkage are complex and influenced by everything from our daily activities to our genetic makeup.
As we age, our bodies exhibit a reduced capacity to construct new muscle protein, even in the presence of anabolic stimuli like resistance exercise or protein intake. This perplexing state has captivated scientists for decades, yet the precise molecular mechanisms behind anabolic resistance have remained shrouded in ambiguity.
However, emerging evidence from the lab of Dr. David J. Glass, MD, sheds light on a potential player in this enigma: the dysregulation of a signaling pathway known as mTOR. By delving into the intricacies of mTOR signaling, Dr. Glass and his team have begun to unravel the mysteries of anabolic resistance, providing insights into the cellular processes that underlie this physiological phenomenon.
To grasp the significance of anabolic resistance, it is essential to comprehend its core concept. Anabolic resistance refers to the diminished ability of the body to construct fresh muscle protein, despite the presence of anabolic stimuli, such as resistance training and protein intake. This phenomenon is intricately intertwined with the process of aging, which brings us closer to the question of why anabolic resistance becomes a prevalent feature as the years pass.
One compelling theory proposes that chronic elevation of mTOR, a key regulator of cellular growth and protein synthesis, plays a pivotal role in the development of anabolic resistance. The idea is that the machinery responsible for controlling cellular size reaches its maximum capacity as we age, rendering us less responsive to protein intake or exercise. In essence, the heightened mTOR activity in aging individuals impedes their ability to further activate mTOR in response to anabolic stimuli—leading to what could be referred to as an mTOR insensitivity to stimuli.
Work conducted in Dr. Glass's laboratory offers compelling support for this hypothesis. Through experiments, his team observed a progressive increase in the basal (fasted) activity of RPS6, a downstream target of mTOR, across the lifespan—indicating an increase in mTOR activity with age. This linear rise in mTOR activity as we age accelerates cellular dysfunction and inflicts damage upon our delicate tissues.
The age-related increase in mTOR signaling coincided with a decrease in muscle mass. Though muscle loss at that age is not a surprise, the coincidence of this loss with the elevated mTOR activation was quite unexpected, given that it favors muscle growth and hypertrophy.
The same study administered rapamycin, a pharmacological agent known to inhibit mTOR, for a duration of six weeks. The outcomes were fascinating. The rapamycin treatment led to a restoration of mTOR signaling intermediates and a reversal of sarcopenia—the harmful loss of muscle mass, strength, and function that afflicts many older adults.
Sarcopenia, a gradual and progressive condition that primarily emerges in middle age, exacerbates with time, leaving individuals more susceptible to frailty and diminished quality of life. By attenuating mTOR activity to "youthful" levels through the use of rapamycin, it seems that we may be able to mitigate the effects of sarcopenia and rekindle the body's muscle-building capabilities.
Rapamycin and Changes in Muscle Tissue Health and Morphology
The research team also examined how rapamycin affected the appearance and health of the muscle tissue. The researchers used different doses of rapamycin, referred to as LD (low dose) and HD (high dose), to see which was more effective. The low-dose treatment turned out to be the key. It led to increased muscle mass in aged rats and more healthy muscle morphology, with other positive molecular changes supporting this growth.
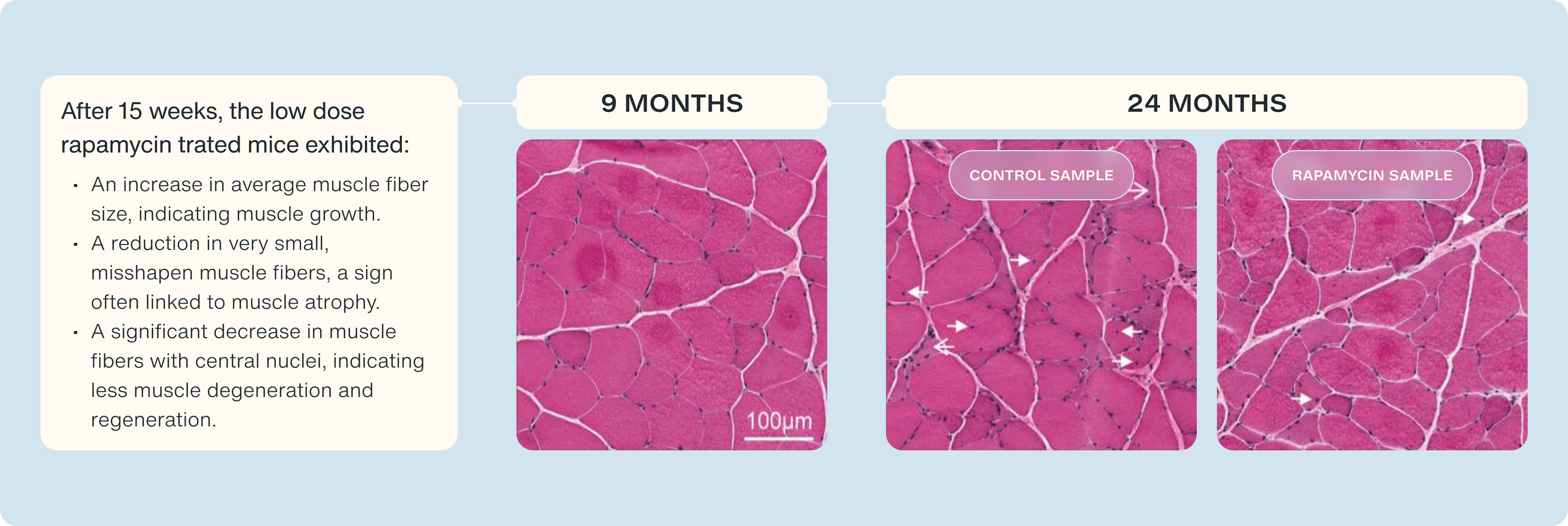
To understand these changes, the scientists performed detailed analyses of the muscle tissue, using techniques like staining with hematoxylin and eosin (H&E). This allowed them to visualize the muscle fibers' structure, comparing healthy young rats with older, sarcopenic ones. They used histology samples of 9 month-year-old rats as an example of healthy tissue, and compared them to the samples 24 month-year-old muscle samples to see the progression of aging on and off of rapamycin.
What did they find?
- Healthy Muscle: Tissue from 9-month-old rats showed normal, healthy muscle characteristics.
- Distressed Muscle: In older rats not treated with rapamycin, they detected signs of distressed and degenerated muscle.
- Improvement with rapamycin: In the low-dose RAD001-treated rats, they observed:
- An increase in average muscle fiber size, indicating muscle growth.
- A reduction in very small, misshapen muscle fibers, a sign often linked to muscle atrophy.
- A significant decrease in muscle fibers with central nuclei, indicating less muscle degeneration and regeneration.
The high-dose rapamycin treatment did not provide these benefits, emphasizing the importance of finding the correct dose.
Conclusions on the Glass Study on Rapamycin and Muscle Growth and Maintenance
These discoveries present a potential avenue for interventions that could combat anabolic resistance and age-related muscle loss. By strategically modulating mTOR activity through pharmacological therapies like rapamycin, it may be possible to restore the body's capacity to respond to anabolic stimuli effectively.
Much work remains to be done before we fully comprehend the complexities of anabolic resistance and the intricate web of mTOR signaling. Dr. Glass's research represents a crucial stepping stone in our desire to unravel the mechanisms underpinning this phenomenon. As we delve deeper into the mysteries of anabolic resistance, we inch closer to a future where aging no longer dictates the deterioration of our muscles but rather opens the door to targeted interventions that sustain our cellular and tissue function over time.